Gonzalo Silva-Aguayo
Agronomist, MS
Facultad de Agronomía, Universidad de Concepción
Avenida Vicente Méndez 595, Chillán CHILE
Introduction
Plants have evolved for over 400 million years and to defend themselves from insect attack they have developed protection mechanisms such as repellents and even insecticidal effects. The oldest known pest control method is human sacrifice, but since it is not effective, or maybe because of the lack of volunteers, dusts began being used and also plant extracts which are mentioned even in the Bible. Massive use of these insecticides has had a long and difficult road because the earliest data gathering done by researchers among farmers and natives revealed a lot of practices based on superstition, which, when tested by scientific methods. were not shown to be effective. After the Second World War the few plant and plant extracts that had shown promising effects, and were of widespread use, were replaced by synthetic insecticides. When synthetic insecticides appeared in the 1940’s some people thought that botanical insecticides would disappear forever but problems like environmental contamination, residues in food and feed and pest resistance brought them back to the fore. There is no doubt botanical insecticides are an interesting alternative to insect pest control, and on the other hand only a few of the more than 250,000 plant species on our planet have been properly evaluated for this purpose. This means the potential for the future may be huge. In fact, plants like neem (Azadirachta indica J., Meliaceae), have shown excellent results and there already are commercial products in the market made from it. But one should not think success is at hand and botanical insecticides will replace all synthetic products. They are only alternatives that may be used in Integrated Pest Management programs and they should be used together with other available control measures.
Brief historical review
Use of plant extracts and powdered plant parts as insecticides goes back at least as far as the Roman Empire. For instance, there are reports that in 400 B.C. during Persian king Xerxes’ reign, the delousing procedure for children was with a powder obtained from the dry flowers of a plant known as pyrethrum (Tanacetum cinerariaefolium, Compositae). The first botanical insecticide, used as such, dates back to the XVII Century when it was shown that nicotine, obtained from tobacco leaves, would kill plum beetles. Around 1850 a new plant insecticide known as rotenone was introduced. It is obtained from the roots of plants called timbó. Up to that time this plant was used for fishing purposes only as natives had known for a long time that throwing root pieces to the water caused fish to start floating a few minutes later, making them very easy to catch. Later on plants with irritating properties like incense and sabadilla were used: extracts from the latter plant were used as decongestants. These plants did not kill insects directly but it was said that they “scared them off.” More recently other plants used are quasia (Quaisa amara, Simaroubaceae), neem or Margosa (A. indica) mentioned above, which besides giving excellent results for insect control are also a source of compounds used against cancer.
In México and several Central American countries even today it is common practice to treat pests with plants known for their insecticidal properties as far back as the era of the Aztecs and Mayans. A case in point is the use of a mixture of corn and beans with chili peppers (Capsicum frutescens; Solanaceae), rue (Ruta graveolens; Rutaceae) or garlic (Allium cepa; Alliaceae).
At the present time there are a number of botanical insecticides being marketed which are extracted from neem, grapefruit seeds and garlic, among other plants. Besides, there are the synthetic copies of natural active ingredients like neonicotinoids where imidacloprid stands out. Lastly, it is worth mentioning that this is a field where new discoveries are made every day. One example is the development of a new kind of insecticide obtained from the plant of Chilean Andes origin known as Calceolaria andina (Scrophulariaceae).
Nature of the compounds
Plants are like natural laboratories where a great number of chemicals are biosynthesized and in fact they may be considered the most important source of chemical compounds there are. Primary plant metabolism synthesizes essential compounds which are present in all plant species. On the other hand, the end products of secondary metabolism are neither essential nor universally present in all plants. Common among these metabolites are compounds with protective action against insects, such as alkaloids, non-proteic amino acids, steroids, phenols, flavonoids, glycosids, glucosinolates, quinones, tanins and terpenoids. Some people support the idea that these compounds do not have a clearly known role, and have even classified them as “metabolic garbage.” However, other researchers point out that those products are important chemical signals in the ecosystem. There is a great deal of variation as far as the concentration of secondary compounds that individuals of a population can express. Besides, there is neither a pattern of maximum yield nor particular storage organs for secondary metabolites, even though it is common that the highest concentrations of these kinds of compounds are found in flowers and seeds.
How do they work?
By definition, an insecticide is a substance that performs a biocidal action on insects due to the nature of its chemical structure. For example, if we kill an insect for our insect museum using a cyanide killing jar we can say that cyanide has an insecticidal effect. However, we cannot say the same about water if raindrops kill aphids, since that mortality cannot be attributed to water’s chemical structure or properties.
Most plant species used for plant protection exhibit an insect deterrent rather than insecticidal effect. This means that in some way those compounds inhibit normal development in insects. This can be done in different ways that will be briefly described below.
Insect Growth Regulators
The growth regulator effect may be seen in several ways. First are those molecules inhibiting metamorphosis, in other words, compounds preventing metamorphosis from taking place at the right time. Other compounds force the insect to go through an early metamorphosis, so that development takes place at a time not favorable for the insect. Lastly, it has been observed that certain molecules may alter hormones related to this function so that insects suffer malformations, are sterile, or die. In fact, one of the most common entomological anecdotes tells that an experiment was being carried out simultaneously in the United States and Hungary. In the latter country the insects went through an additional immature stage before molting to adults. All methods were reviewed but no difference was found until the paper towels used to provide water for the insects were analyzed and then it was found that in the two countries the paper towels were made from different trees. In Europe they used Abies balsamea, a coniferous tree, very common in Hungary, which contains a plant hormone which induced the additional molt. A more practical example is the case of basil plant (Ocimum basilicum) from which the compound juvocineme II is extracted and which later on served as the model for synthesis of piriproxifen and fenoxicarb.
Feeding deterrents
Feeding deterrence is perhaps the most studied mode of action for plant derivatives used for insect pest management. Strictly speaking, a feeding deterrent is a compound that once probed by the insect, causes it to stop feeding and starve to death. Many compounds showing this activity are terpenes and most have been isolated from medicinal plants native to Africa and India.
Repellents
The use of plants as repellents is very old but has not received the necessary attention for proper development. Use has been achieved with compounds having bad odor or irritant effects as is the case for garlic and hot peppers. A clear example can be seen in uses by Guatemalan and Costa Rican natives who used to "paint" containers or sprinkle them with garlic powder before using them to store corn or beans to prevent the grain from being damaged by weevils (weevil punctures) and also to shoo away rodents. Lastly, it is not uncommon to hear of home recipes mentioning the use of fennel (Foniculum vulgare), rue (Ruta graveolens) and eucalyptus (Eucaliptus globolus) among other aromatic plants to repel clothes moths.
Confusants
Chemical compounds in a given plant are unequivocal signs for insects to find their food source. In fact, there are cases like the monarch butterfly, which feeds on plants that are highly poisonous to other organisms, but that are found by the butterfly because of the poisonous substance. One way to use this characteristic in IPM has been setting up traps and either spraying them with infusions of certain plants that are more attractive to the insect or of the same plant but from other areas so that the insect will have so many sources of stimuli that they will not be able to find the plant we wish to protect. Another option is to set up traps containing watery extracts of the plant so that insects will “land” in the traps and not on the crop.
So, thinking about the above mentioned information we should consider all those compounds which we know have insect deterrents and have preventive rather than curative effects.
Advantages and Disadvantages of Botanical Insecticides
Advantages
- Plants producing the above mentioned compounds are known by the farmer because most of the time they grow in the same general area.
- Often these plants also have other uses like household insect repellents or are plants with medicinal applications.
- The rapid degradation of the active product may be convenient as it reduces the risk of residues on food.
- Some of these products may be used shortly before harvesting.
- Many of these products act very quickly inhibiting insect feeding even though long term they do (will?) not cause insect death.
- Since most of these products have a stomach action and are rapidly decomposed they may be more selective to insect pests and less aggressive with natural enemies.
- Most of these compounds are not phytotoxic.
- Resistance to these compounds is not developed as quickly as with synthetic insecticides.
Disadvantages
- Most of these products are not truly insecticides since many are merely insect deterrents and their effect is slow.
- They are rapidly degraded by UV light so that their residual action is short.
- Not all plant insecticides are less toxic to other animals than the synthetic ones.
- They are not necessarily available season long.
- Most of them have no established residue tolerances.
- There are no legal registrations establishing their use.
- Not all recommendations followed by growers have been scientifically verified.
Which plants to use?
There are a lot of publications with lists of plants with insecticidal properties. For example, in 1950, Heal et al. reported approximately 2,500 plants in 247 families with some sort of toxic property against insects. But to use them it is not enough that the plant be considered as promising or even with proven insecticidal properties. It is also necessary to conduct an analysis of the risks to the environment and to human health. Another example is that it may not be appropriate to recommend the use of plants in danger of extinction, that are difficult to find, or if their use results in important alterations to their population density in natural conditions. For example, if tomorrow it is discovered that baoba wood has a compound which may be an insecticide, that does not mean we are going to cut the baoba trees. For these reasons and with the purpose of obtaining maximum benefit from a plant with insecticidal properties, but without degrading the ecosystem, the properties that an ideal insecticidal plant should have are listed below (Silva, 2001):
- It should be a perennial.
- It should have a wide distribution and be present in large numbers in nature. Otherwise it should be possible to be grown by agricultural procedures.
- The plant parts to be used should be removable: leaves, flowers or fruit.
- Harvesting should not mean destruction of the plant (avoid the use of roots or bark).
- The plants should require small space, reduced management and little water and fertilization.
- The plant should have additional uses (for example medicinal use).
- The plant should not otherwise have a high economic value.
- The active ingredient should be effective at low rates.
If it is natural, is it not poisonous?
It would be a big mistake to consider products of plant origin, and this includes botanical insecticides, harmless merely because they are natural. There are large numbers of plant products which are highly toxic; remember that ancient history mentions that Socrates was sentenced to death by drinking a diluted extract of hemlock (Cicuta spp.). Schmutz and Breazeale (1986), in their book “Plants that Poison” point out around 120 species of plants containing substances that are toxic to humans, including plants as common as almond trees, some bean species, garlic, strawberries and apples, among others. So we must not forget that the toxic potential of a molecule is the nature of its chemical structure and not its origin. As Paracelsus wrote in 1564 “the difference between something that kills and something that cures is the rate.”
Laboratory Copies
Plants have been used not only directly as insecticides but also as a source for a series of synthetic insecticides developed in the laboratory. From the pest control point of view. one problem is that botanical insecticides disappear soon after application because they are decomposed by the action of light and temperature so that they remain on the plant only briefly (typically no more than 24 hours). This has prompted industry to modify molecules found in plants so that they are more toxic and more persistent.. These improvements offer the advantage that the crop does not have to be sprayed virtually every day and that there will be less pressure to harvest the insecticidal plants into extinction. A clear example of this may be found in two insecticide families of widespread use in agriculture, medicine and urban environments: pyrethroids and carbamates which are synthetic derivatives of molecules isolated from plants like pyrethrum (T. cinerariaefolium) and Calabar beans (Physostigma venenosum), respectively.
Description of Some Compounds
Rotenone
Rotenone is a flavonoid extracted from the roots of two plants: Derris spp. (Fabaceae) and Lonchocarpus spp. (Fabaceae). The first one gives up to 13% of rotenone while the second only about 5%. Derris spp. is a native to Eastern tropics, while Lonchocarpus spp. is native to western hemisphere. Rotenone is a contact and ingestion compound, which acts as a repellent too. Its mode of action involves the inhibition of the electron transport at the mitochondrial level, thus blocking phosphorylation of ADP to ATPthereby inhibiting insect metabolism. Insects poisoned with Rotenone show the following symptoms of intoxication: a drop in oxygen consumption, respiratory depression and ataxia leading to convulsions and finally to paralysis and death by respiratory arrest.
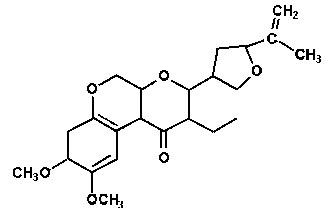
Sabadilla
This is a compound that can be obtained from seeds of a plant of South American origin known as Schoenocaulon officinale (Liliaceae). Seeds of this plant have been shown to have high concentrations of alkaloids which impart its toxic properties. The mode of action is disruption of neuron cell membranes causing reduction of nerve activity, paralysis and death. The ground seeds are one of the plant insecticides with the lowest mammal toxicity but that is not the case with their isolated alkaloids which are both highly toxic and skin irritants.
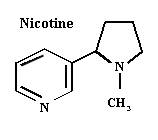
Nicotine
Nicotine is an alkaloid obtained from some plants in the Solanaceae family, particularly tobacco (Nicotiana tabacum). The insecticidal properties of nicotine were recognized in the first half of the XVI century. This compound is not found free in the plant but in the form of maleates and citrates. Nicotine is essentially a non-persistent contact insecticide. Its mode of action consists in mimicking acetylcholine when it binds with its receptor in the post-synaptic membrane of the muscular union. The acetylcholinic receptor, is a site of action of the postsynaptic membrane which reacts with acetylcholine and alters the membrane permeability. Nicotine activity causes the production of new nerve impulses which cause convulsions, and death. An important new class of insecticides, commonly known as neo-nicotinoids, are synthetic copies or derivatives of the nicotine structure. These include imidacloprid, thiacloprid, nitempiram, acetamiprid and thiamethoxam, among others.
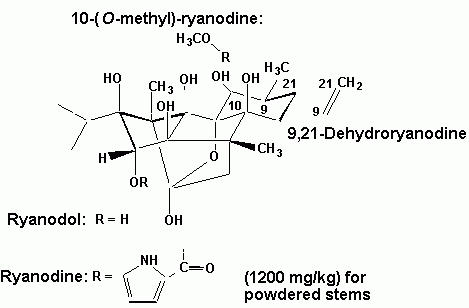
Ryania
This compound is obtained from the roots and stems of a plant native to South America known as Ryania speciosa (Flacourtiaceae). From this plant may be obtained a series of alkaloids, of which the most important is ryanodina. This alkaloid is effective as a contact or stomach poison and directly prevents muscles from contraction,causing paralysis.
Azadiractin
This compound is a tetraterpenoid characteristic of the Meliaceae family but particularly from the Neem tree (A. indica), indigenous to India. The compound is found in bark, leaves and fruits of the tree but seeds have the highest concentration. This compound has not yet been synthesized in the laboratory, but when isolated and tested pure the results have been less than when extracts are used. In the extract 18 compounds have been identified among which salanine, meliantrol and azadiractin are most prominent, the latter being in the highest concentration. Azadirachtin shows antifeedant activity, is a growth regulator, inhibits oviposition and is also a sterilizing compound. Today, commercial formulations of neem may be found with names like Neem Gold, Neemazal, Econeem, Neemark, Neemcure and Azatin among others, in many countries including the United States, India, Germany and several Latin American countries.
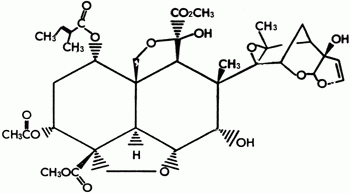
Pyrethrins
Pyrethrins, which are esters with insecticidal properties, are obtained from pyrethrum (T. cinerariaefolium) flowers. The compounds obtained from this plant which have known insecticidal activity are six esters formed by the combination of the acids chrysanthemic and pyrethric and the alcohols piretrolone, cinerolone and jasmolone. These compounds act both on the central nervous system and in the peripheral nervous system causing repetitive discharges, followed by convulsions. Research has shown that these compounds block sodium ion influx resulting in the channels being affected by intermolecular forces which causes alterations in moving ion conductivity as a result of tchanges in the channels. There is no doubt that the most important characteristic of these compounds is their irritating effect or “knock down” which causes the insect to stop feeding as soon as it encounters a treated surface. Pyrethrins are the best example of products copied in the laboratory since their modifications gave rise to the pyrethroid family of insecticides.
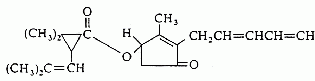
Resistance
Most insecticides of botanical origin are extracts containing a group of active ingredients of diverse chemical nature. From a resistance point of view the short residual life of plant insecticides may be considered as a positive since there will be a very low probability that two extracts would always be identical so that selective pressure on the pest species will not always be the same. Even if all the same compounds are found in the extract, concentrations almost always will be different. Generally speaking, insect resistance takes longer to develop to a mixture of natural active compounds than to any one individual component. This may be because it is more difficult to detoxify a compound complex than a single molecule. In a lab evaluation with green peach aphid, Myzus persicae, when azadirachtin was applied alone, the insect was able to develop a level of resistance nine times higher than in the initial population within 35 generations. Using the whole neem extract (containing the same azadirachtin concentration) no indication of resistance was seen in the same time period.
Future Perspective
The main market for plant origin insecticides is in parks and gardens. This is due to their low environmental persistence so that people will be less exposed to the toxic compounds. It is expected that within 10-15 years, these compounds will increase 25% in insecticide market share and they will not be limited to garden areas but there may be a massive growth towards urban and agricultural uses. However, even though plant origin insecticides are advantageous options from an ecological point of view, it would be unreasonable to think they will completely replace synthetic insecticides. On the contrary, it will be quite logical to expect a complementary use and the coexistence of the two kinds of compounds as it happens today with pyrethrum and synthetic pyrethroids in some IPM programs. On the other hand, organic agriculture is a market with high demand for plant origin insecticides as organic growers cannot use conventional agrichemicals. At this time this market is expanding and as a rule rates of return are high, so that this is an important “niche” to take care of. Unfortunately it is commonly thought that organic agriculture is the same as conventional agriculture except that no synthetic agrichemicals are used. This mistake may lead some growers to lose confidence in their objectives if they do not get the expected results and they may hold on to their old preference for synthetic insecticides.
Plant origin insecticides also have a great advantage by being compatible with other low risk options which are acceptable for insect management, such as pheromones, oils, detergents, entomopathogenic fungi, predators and parasitoids, among others, which greatly increase probabilities of being integrated in IPM programs. Lastly, it may be pointed out that, short term, there is no doubt that new plants will be studied, as is the case of Calceolaria andina. New technologies will be developed and perfected to isolate and identify molecules. Extension and education programs on plant origin insecticides will increase and improve and new sources of raw materials will be found for their manufacture, as it is happening today with discarded citrus peels in the United States. Quite probably registration procedures will be faster in developing countries.
References
- Addor, R. W. 1995. Insecticides. In: Godfrey C.R.A. (Ed) Agrochemicals from natural products. Marcel Dekker, Inc. pp 1-62.
- Arnason, J. T., B. J. Philogene and P. Morand (EDS). 1989. Insecticides of plant origin. American Chemical Society, Washington, DC. USA. 213 p.
- Coats, J. R. 1994. Risks from natural versus synthetic insecticides. Annu. Rev. Entomol. 39:489-515.
- Heal, R., E. Rogers, R. T. Wallace y O. Starnes. 1950. A survey of plants for insecticidal activity. Lloydia 13(2):89-162.
- Rosenthal, G., and M. Berenbaum (Eds). 1991. Herbivores: Their Interactions with secondary plant metabolites. Academic Press, Inc. San Diego. USA. 467 p.
- Schmutz, E. y L. Breazeale.1986. Plants that Poison. Northland Press. Flagstaff. Arizona. USA. 241 p.
- Silva, G., A. Lagunes, J. C. Rodríguez y D. Rodríguez. 2002. Insecticidas vegetales; Una vieja-nueva alternativa en el control de plagas. Revista Manejo Integrado de Plagas (CATIE) (en prensa).