Douglas A. Landis
Department of Entomology
Michigan State University
East Lansing, MI 48824
David B. Orr
Department of Entomology
North Carolina State University
Method Unit II, 840 Method Rd.
Raleigh, NC 27607
e-mail: [email protected]
Introduction
In recent decades, elevated awareness of the impacts of pesticide use on the environment and human health have resulted in efforts to reduce reliance on chemical controls. Many countries have instituted more stringent regulation of pesticide manufacture, registration and use, thereby increasing the cost, and decreasing the availability of these tools. In many cases, the pests themselves have indicated the need for change, with pesticide resistance now a common reality in many weeds, insects and diseases. The need for alternatives to pesticides is clear, but where will these solutions come from? A recent report by the U. S. Congress, Office of Technology Assessment (U. S. Congress, OTA 1995) indicates that biologically based technologies such as biological control could be more widely used to solve pressing needs in pest management.
The use of natural enemies to reduce the impacts of pests has a long history. The ancient Chinese, observing that ants were effective predators of many citrus pests, augmented their populations by taking their nests from surrounding habitats and placing them into their orchards. Today's insectaries and air-freight delivery of natural enemies across the country or around the world are simply modern adaptations of these original ideas. In this article we will examine approaches to biological control and applications of these approaches in modern pest management. While the principles of biological control can be applied against various pest organisms (e.g. weeds, plant pathogens, vertebrates and insects), we will limit our discussion to the use of biological control of insects, primarily using other insects as natural enemies.
Approaches to Biological Control
There are three general approaches to biological control; importation, augmentation and conservation of natural enemies. Each of these techniques can be used either alone or in combination in a biological control program.
Importation
Importation of natural enemies, sometimes referred to as classical biological control, is used when a pest of exotic origin is the target of the biocontrol program. Pests are constantly being imported into countries where they are not native, either accidentally, or in some cases, intentionally. Many of these introductions do not result in establishment or if they do, the organism may not become pests. However, it is not uncommon for some of these introduced organisms to become pests, due to a lack of natural enemies to suppress their populations. In these cases, importation of natural enemies can be highly effective (Caltagirone 1981).
Once the country of origin of the pest is determined, exploration in the native region can be conducted to search for promising natural enemies. If such enemies are identified, they may be evaluated for potential impact on the pest organism in the native country or alternatively imported into the new country for further study. Natural enemies are imported into the US only under permit by the US Department of Agriculture. They must first be placed in quarantine for one or more generations to be sure that no undesirable species are accidentally imported (diseases, hyperparasitoids etc.). Additional permits are required for interstate shipment and field release.

Biological control of the alfalfa weevil, Hypera postica (Gyllenhall) is a example of a successful program using importation of natural enemies (Bryan et al. 1993). The alfalfa weevil, a native of Europe, was originally detected in the US in Utah in 1904. A second introduction was detected on the East coast in 1951. By 1970, the weevil had spread to all 48 contiguous states and become a serious pest of alfalfa. Some importation's of natural enemies began as early as 1911, however, a major program aimed at biological control of the weevil was initiated in 1957. In this program, USDA ARS personnel conducted foreign exploration in Europe resulting in the eventual importation of 12 parasitoid species. Six of these species became established and are credited with contributing to the reduction in the weevil's pest status in the eastern US (Day 1981).
Augmentation
Augmentation is the direct manipulation of natural enemies to increase their effectiveness. This can be accomplished by one, or both, of two general methods: mass production and periodic colonization; or genetic enhancement of natural enemies. The most commonly used of these approaches is the first, in which natural enemies are produced in insectaries, then released either inoculatively or inundatively. For example, in areas where a particular natural enemy cannot overwinter, an inoculative release each spring may allow the population to establish and adequately control a pest. Inundative releases involve the release of large numbers of a natural enemy such that their population completely overwhelms that of the pest. Augmentation is used where populations of a natural enemy are not present or cannot respond quickly enough to the pest population. Therefore, augmentation usually does not provide permanent suppression of pests, as may occur with importation or conservation methods.
An example of the inoculative release method is the use of the parasitoid wasp, Encarsia formosa Gahan, to suppress populations of the greenhouse whitefly, Trialeurodes vaporariorum (Westwood), (Hussey & Scopes 1985, Parrella 1990). The greenhouse whitefly is a ubiquitous pest of vegetable and floriculture crops that is notoriously difficult to manage, even with pesticides. Releases of relatively low densities (typically 0.25 to 2 per plant, depending on the crop) of E. formosa immediately after the first whiteflies are detected on a greenhouse crop can effectively prevent populations from developing to damaging levels. However, releases should be made within the context of an integrated crop management program that takes into account the low tolerance of the parasitoids to pesticides.
Conservation
In any biological control effort, conservation of natural enemies is a critical component. This involves identifying the factor(s) which may limit the effectiveness of a particular natural enemy and modifying them to increase the effectiveness of the beneficial species. In general, conservation of natural enemies involves either, reducing factors which interfere with natural enemies or providing resources that natural enemies need in their environment.
Many factors can interfere with the effectiveness of a natural enemy. Pesticide applications may directly kill natural enemies or have indirect effects through reduction in the numbers or availability of hosts. Various cultural practices such as tillage or burning of crop debris can kill natural enemies or make the crop habitat unsuitable. In orchards, repeated tillage may create dust deposits on leaves, killing small predators and parasites and causing increases in certain insect and mite pests. In one study, periodic washing of citrus tree foliage resulted in increased biological control of California red scale, Aonidiella aurantii (Maskell) due to increased parasitoid efficiency (Debach & Rosen 1991). Finally, host plant effects such as chemical defenses which are harmful to natural enemies but to which the pest is adapted, can reduce the effectiveness of biological control. Some pests are able to sequester toxic components of their host plant and use them as defense against their own enemies. In other cases, physical characteristics of the host plant such as leaf hairiness, may reduce the ability of the natural enemy to find and attack hosts.
Ensuring that the ecological requirements of the natural enemy are met in the cropping environment is the other major means of conserving natural enemies. To be effective, natural enemies may need access to; alternate hosts, adult food resources, overwintering habitats, constant food supply, and appropriate microclimates (Rabb et al. 1976). In a classic example, Doutt & Nakata (1973) determined that Anagrus epos Girault, principal parasitoid of the grape leafhopper, Erythroneura elegantula Osborne in California grape vineyards required an alternate host for overwintering. This host, another leafhopper, only overwintered on blackberry foliage in riparian areas, often quite distant from the vineyards. Vineyards close to natural blackberry stands experienced earlier colonization by the parasitoid in the spring and better biological control. Wilson et al. (1989) found that French prune trees which harbor another overwintering host, could be planted upwind of vineyards and effectively conserve Anagrus epos.
Current Applications of Biological Control
Biological control is an exciting science because it constantly incorporates new knowledge and techniques. In this section we will illustrate several ways in which time honored approaches to biological control are being adapted to meet today's pest management challenges.
Modern Approaches in Augmentation of Natural Enemies
Because most augmentation involves mass-production and periodic colonization of natural enemies, this type of biological control has lent itself to commercial development. There are hundreds of biological control products available commercially for dozens of pest invertebrates, vertebrates, weeds, and plant pathogens (Anonymous 1995).
The practice of augmentation differs from importation and conservation in that making permanent changes in a agroecosystem to improve biological control is not the primary goal. Rather, augmentation generally seeks to adapt natural enemies to fit into existing production systems. For example, cultures of the predatory mite, Metaseiulus occidentalis (Nesbitt) were laboratory-selected for resistance to pesticides commonly used in an integrated mite management program in California almond orchards (Hoy 1985). This program has saved growers $24 to $44 per acre per year in reduced pesticide use and yield loss (Headley & Hoy 1987). Genetic improvement of several predators and parasitoids has been accomplished with traditional selection methods (Hoy 1992), and appears possible with recombinant DNA technology.
An excellent example of an augmentative practice than has been successfully adapted to a wide variety of agricultural systems is the inundative release of Trichogramma wasps. These minute endoparasitoids of insect eggs are released in crops or forests in large numbers (up to several million/ha) timed to the presence of pest eggs. Trichogramma are the most widely augmented species of natural enemy, having been mass-produced and field released for almost 70 years in biological control efforts. Worldwide, over 32 million ha of agricultural crops and forests are treated annually with Trichogramma spp. in 19 countries, mostly in China and republics of the former Soviet Union (Li 1994).
In China, agricultural production and pest management systems capitalize on low labor costs, and generally follow highly innovative yet technologically simple processes. For example, Trichogramma spp. that are inundatively released to suppress sugarcane borer, Chilo spp., populations in sugarcane are protected from rain and predators inside emergence packets. Insectary-reared parasitized eggs are wrapped in sections of leaves which are then slipped by hand over blades of sugarcane. Most Trichogramma production in China takes place in facilities producing material for a localized area. These facilities range from open air insectaries to mechanized facilities that are leading the world in development of artificial host eggs.
One of the barriers to wider implementation of biological control in western agriculture has been socio-economics (van Lenteren 1990). In current large-scale production agricultural systems, a premium is placed on efficiency and economy of scale. Entire support industries have developed around the application of agrichemicals, including application equipment manufacturing, distribution and sales, as well as application services. In order for biological control products to not be at odds with these industries, and to compete strongly with pesticides, they should have many of the same characteristics. Ideally, they should be as effective as pesticides, have residual activity, be easy to use, and they should have the capacity to be applied quickly on a large scale with conventional application equipment.
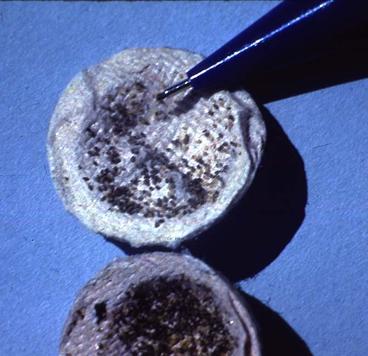
In Western Europe, almost two decades of intensive research resulted in the commercial marketing of three products utilizing the European native, Trichogramma brassicae Bezdenko, to suppress the European corn borer, Ostrina nubilalis Hübner, in corn fields (Bigler et al. 1989). These products are annually applied to approximately 7,000 ha in each of Switzerland and Germany, 150 ha in Austria, and 15,000 ha in France. All three products are based on manufactured plastic or paper packets designed to provide protection for the wasps against weather extremes and predation until emergence in the field.
As in the Chinese example above, European Trichogramma products are for the most part applied to crop fields by hand. One exception is the product called, Trichocaps which can be broadcast either by hand or by aircraft using conventional application equipment. Trichocaps packets are actually hollow walnut-shaped cardboard capsules (2 cm. diam.) that each contain approximately 500 parasitized Mediterranean flour moth, Ephestia kuehniella Zwolfer, eggs (Kabiri et al. 1990). Developing Trichogramma inside capsules are induced into an overwintering (diapause) state in the insectary, then stored in refrigerated conditions for up to nine months without loss of quality. This system allows for production of product during winter months, then distribution to growers when needed in the summer.
Once removed from cold storage, Trichogramma inside the capsules will begin development and begin emergence approximately 100 Celsius degree days later. This 'reactivation' process can be manipulated so that capsules containing Trichogramma at different developmental stages can be applied to fields at the same time, extending the emergence period of parasitoids and increasing the 'residual' activity of a single application to approximately one week. Planning and preparation of the product for application is done by the company so that growers are only responsible for applying the product to crop fields.
Cooperative research over the last 5 years (between BIOTOP, Pioneer Hi-Bred Intl., BASF, Univ. of Illinois, Iowa State University, Michigan State University, Purdue University, and Pest Management Co. of Nebraska) has resulted in successful commercial-scale pilot testing of this method in North America on seed corn and field corn production systems (Orr 1993, Orr et al. unpublished data). This strategy now has the potential for immediate commercial implementation in North America.
Landscape Ecology and the Conservation of Natural Enemies
The study of disturbance and its effects on community dynamics and the emergence of the discipline of landscape ecology are impacting the way we think about the conservation of natural enemies. Over the past 15 years, ecologists have come to recognize the central role that disturbance plays in the structuring of ecological communities (Pickett & White 1985, Reice 1994). While the most highly disturbed terrestrial ecosystems may have one disturbance event every several years (e.g. fire in grasslands), many agricultural ecosystems experience multiple events per growing season (plowing, planting, nutrient and pesticide applications, cultivation and harvest). From an ecological point of view, the outcomes are predictable (Odum 1985). Highly disturbed systems exhibit decreased species diversity and shortened food chains, resulting in the few well adapted species (i.e. pests) having few natural enemies to suppress their populations. This requires that additional disturbance events be initiated (i.e. pesticide applications) which, while controlling the initial negative symptom, may precipitate its reoccurrence.
Current systems of crop production also shape the physical structure of our agricultural landscapes (Forman & Godron 1986). With increased reliance on mechanization and pesticides, diversity in farmlands has rapidly disappeared and the impacts on natural enemies are only now beginning to be understood (Ryszkowski et al. 1993). In general, increased habitat fragmentation, isolation and decreased landscape structural complexity tend to destabilize the biotic interactions which serve to regulate natural ecosystems (Kruess & Tscharntke 1994, Robinson et al. 1992).
The goal of an ecological approach to conservation biological control is to modify the intensity and frequency of disturbance to the point where natural enemies can function effectively. This will need to occur at field, farm and larger landscape-levels. Within fields, modification of tillage intensity and frequency (reduced tillage or no-tillage) can leave more plant residue on the soil surface and have a positive impact on predators (ground beetles and spiders). Intercropping can also modify the microclimate of crop fields making them more favorable for parasitoids.

At the farm level, the presence and distribution of non-crop habitats can frequently be critical to natural enemy survival. Eriborus terebrans (Gravenhorst) is a wasp which parasitizes European corn borer larvae. Female Eriborus require moderate temperatures (<90° F) and a source of sugar (nectar of flowering plants or aphid honeydew). Neither of these conditions is met in a conventionally managed corn field. Therefore, wasps seek more sheltered locations in wooded fencerows and woodlots where they find reduced temperatures, higher relative humidity and abundant sources of adult food. European corn borer larvae in corn field edges near these types of habitats are parasitized at two to three times the rate of those in field interiors (up to 40%) (Landis & Haas 1992). Current research is examining the potential of modifying corn production systems by creation of natural enemy resource habitats to provide critical resources and increase natural control of European corn borers. Intercrops, strip crops, as well as modification of grass waterways, shelterbelts, buffer and riparian zones are promising techniques.

Finally, at the landscape-level, the physical structure of agricultural production systems can also influence pest and natural enemy diversity and abundance. In a study contrasting simple versus mosaic landscapes, Ryszkowski et al. (1993) concluded that natural enemies are more dependent on refuge habitats than are pests and the greater abundance of these refuges in the mosaic landscapes resulted in their higher diversity, abundance and ability to respond to prey numbers. Marino and Landis (in press) examined parasitism of true armyworm, Pseudaletia unipuncta (Haworth), in structurally-complex versus simple agricultural landscapes. Overall parasitism in the complex sites was more than three times higher than in the simple sites (13.1% versus 3.4%). Differences were largely attributable to one wasp species, the braconid, Meterous communis (Cresson) which was far more abundant in complex sites. They hypothesized that abundance and proximity of preferred habitats for alternate hosts of M. communis may account for the observed differences.
In the past, conservation was typically attempted one species at a time, concentrating on meeting the needs of what was deemed the most important natural enemy in a particular system. While this will continue to be an enormously useful approach, it now seems possible that basic ecological theory could inform the design and management of landscapes to conserve and enhance the effectiveness of entire communities of natural enemies.
Summary
Importation, augmentation and conservation of natural enemies constitute the three basic approaches to biological control of insects. Specific techniques within these approaches are constantly being developed and adapted to meet the changing needs of pest management. Improvements in rearing and release techniques and genetic improvement of natural enemies have resulted in more effective augmentation programs. Application of new ecological theory is transforming the way we look at conservation of natural enemies. Continued refinement and adaptation of biological control approaches and applications are necessary if the full potential of this biologically based pest management strategy is to be fulfilled.
References Cited
- Bigler, F., S. Bosshart and M. Walburger. 1989. Bisherige und neue Entwicklungen bei der biologischen Bekampfung des Maiszunslers, Ostrinia nubilalis Hbn., mit Trichogramma maidis Pint. et Voeg. in der Schweiz. Landwirtscahft Schweiz Band 2: 37-43.
- Bryan, M. D., R. J. Dysart and T. L. Burger. 1993. Releases of Introduced Parasites of the Alfalfa weevil in the United States, 1957-88. USDA APHIS Misc. Publ. No. 1504. 203 pp.
- Caltigirone, L. E. 1981. Landmark examples in classical biological control. Ann. Rev. Entomol 26:213-32.
- Day, W. H. 1981. Biological control of the alfalfa weevil in the Northeastern United States. Chap 25 In. Papavizas, G. C. ed. Biological Control in Crop Protection. Allanheld, Osmun & Co., Montclair, NJ.
- Debach, P., and D. Rosen. 1991. Biological Control by Natural Enemies. Cambridge Univ. Press, Cambridge, UK. 440 pp.
- Doutt, R. J., and J. Nakata. 1973. The Rubus leafhopper and its egg parasitoid: an endemic biotic system useful in grape pest management. Environ. Entomol. 3:381-6.
- Forman, R. T. T., & M. Godron. 1986. Landscape Ecology. New York: John Wiley and Sons.Headley, J. C. & M. A. Hoy. 1987. Benefit/cost analysis of an integrated mite management program for almonds. J. Econ. Entomol. 80:555-559.
- Hoy, M. A. 1985. Almonds: Integrated mite management for California almond orchards, pp. 299-310. In: W. Helle & M. Sabelis [eds.], Spider mites,world crop pests. Elsevier, Amsterdam.
- Hoy, M. A. 1992. Biological control of arthropods: genetic engineering and environmental risks. Biological Control 2:166-170.
- Hussey, N. W. & N. Scopes. 1985. Biological pest control: the glasshouse experience. Cornell Univ. Press, Ithaca.
- Kabiri, F., J. Frandon, J. Voegele, N. Hawlitzky, & M. Stengel. 1990.[Evolution of a strategy for inundative releases of Trichogramma brassicae Bezd. (Hym Trichogrammatidae) against the European corn borer, Ostrinia nubilalis Hbn. (Lep. Pyralidae)]. In proceedings, ANPP-Second International Conference on Agricultural Pests, Versailles, 4-6 Dec., 1990.
- Kruess, A., & T. Tscharntke. 1994. Habitat fragmentation, species loss, and biological control. Science 264: 1581-1584.
- Li, Li-Ying. 1994. Worldwide use of Trichogramma for biological control on different crops: a survey, pp. 37-54. In: E. Wajnberg & S. A. Hassan, eds. Biological control with egg parasitoids. CAB International, Wallingford.
- Landis, D. A., and M. Haas. 1992. Influence of landscape structure on abundance and within-field distribution of Ostrinia nubilalis Hübner (Lepidoptera: Pyralidae) larval parasitoids in Michigan. Environ. Entomol. 21: 409-416.
- Marino, P. C., & D. A. Landis. Effect of landscape structure on parasitoid diversity and parasitism in agroecosystems. Ecological Applications: in press.
- Odum, E. 1985. Trends expected in stressed ecosystems. BioScience 35(7): 419-421.
- Orr, D. B. 1993. Biological control tactics for European corn borer. Proc. Illinois Crop Prot. Wksp., Mar 3-5, 1993. Champaign, IL.
- Parrella, M. L. 1990. Biological pest control in ornamentals: status and perspectives. SROP/WPRS Bull. XIII/5:161-168.
- Pickett, S. T. A., & P. S. White, eds. 1985. The ecology of natural disturbance and patch dynamics. San Diego: Academic Press.
- Reice, S. R. 1994. Nonequilibrium determinants of biological community structure. Am. Scientist 82: 424-435.
- Robinson, G. R., R. D. Holt, M. S. Gaines, S. P. Hamburg, M. L. Johnson, H. S. Fitch, & E. A. Martinko. 1992. Diverse and contrasting effects of habitat fragmentation. Science 257: 524-526.
- Ryszkowski, L., J. Karg, G. Margarit, M. G. Paoletti, & R. Zlotin. 1993. Above-ground insect biomass in agricultural landscapes of Europe. In Landscape ecology and agroecosystems, ed. R. G. H. Bunce, L. Ryszkowski, & M. G. Paoletti, pp. 71-82. Boca Raton: Lewis Publ.
- U. S. Congress, Office of Technology Assessment. 1995. Biologically Based Technologies for Pest Control. OTA-ENV-636. U.S. Government Printing Office, Washington, DC. 204 pp.
- van Lenteren, J. C. 1988. Implementation of Biological Control. Am. J. Alternative Agric. 3: 102-109.
- Wilson, L. T., C. H. Pickett, D. L. Flaherty and T. A. Bates. 1989. French prune trees: refuge for grape leafhopper parasite. Calif. Agric. 43(2):7-8.
Key References
- Anonymous. 1995. 1996 Directory of least-toxic pest control products. IPM Practitioner 17 (11/12): 1-48. (This reference manual provides a complete listing of the biological control products available worldwide, and can be ordered for $10 from: BIRC, P.O. Box 7414, Berkeley, CA 94707).
- Rabb, R. L., R. E. Stinner and R. van den Bosch. 1976. Conservation and augmentation of natural enemies. pp. 233-54 In. Theory and Practice of Biological Control. C. B. Huffaker and P. S. Messenger eds. Academic Press, New York, NY.