Jeffrey R. Bloomquist
Neurotoxicology Laboratory, Department of Entomology and Nematology
Emerging Pathogens Institute
2055 Mowry Road, Box 100009
University of Florida, Gainesville, FL, 32610
Phone: (352) 273-9417
Fax: (352) 273-9420
Introduction and Scope
This chapter focuses on the chemistry and mode of action of insecticides, and the term “insecticide” will encompass both organic natural products and synthetic compounds. Discussion of synthetic insecticides will emphasize the major classes of commercial materials. Representative chemical structures will be given, especially those that illustrate important structure-toxicity relationships of commercial molecules. No attempt is made to include all the important compounds within a group, and no endorsement of, or discrimination against, any compound is either stated or implied by its inclusion or omission in this chapter. Compounds with unknown or poorly defined modes of action are omitted, as well as chemical classes having only a few members or narrow species selectivity. Emphasis is placed on compounds registered for use, but older materials are sometimes shown for historical perspective and to illustrate important principles. Natural products will be discussed because many of these materials have advantageous properties for integrated pest management and also because the use of these materials in “organic” pest control is increasing. Discussion of insecticide mode of action will focus on interactions with protein targets and the resulting expression of toxicity in the insect. Sufficient background information on the affected physiological processes will also be given so that the actions of insecticides on these processes can be clearly understood. This chapter will not cover genetically engineered plants or baculoviruses expressing Bacillus thuringiensis toxins or other polypeptides, nor chemicals that are used in pest management because of their nonlethal (e.g., pheromones, anti-feedants, synergists, etc.) or non-specific (e.g., insecticidal oils or soaps, etc.) properties.
The length of the sections on each class of insecticide varies, depending on the number of commercial compounds within a group and the amount of available information on mode of action. Chemical structure and mode of action figures are highlighted in italics. Along with each chemical structure is given the available rat oral LD50 (dose that kills 50% of a group of test animals), expressed in mg active ingredient/kg body weight. This information will provide a perspective on the comparative acute toxicity of different compounds. Any material lacking an LD50 value means it was not publicly available at the time this chapter was submitted. At the end of the chapter are a list of the references used in assembling this information, along with some related internet resources on the chemistry and toxicology of insecticides.
Compounds Affecting Voltage-Dependent Sodium Channels
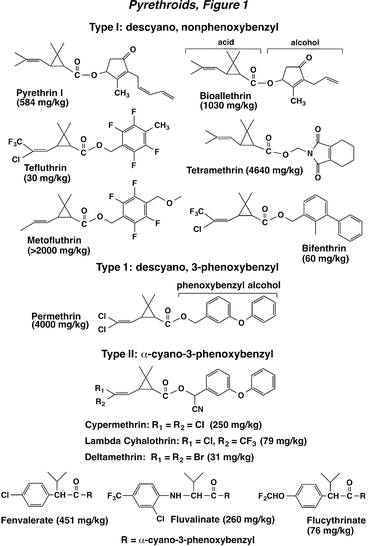
The pyrethroid insecticides are widely used compounds, typically esters of chrysanthemic acid having a high degree of lipophilicity (fat solubility). The original compounds in this series were the natural pyrethrins (e.g., pyrethrin I), which are isolated from the flowers of chrysanthemum. Pyrethroid chemistry and action are classified as Type 1 or Type 2, depending on the alcohol substituent in the molecule. The Type 1 group is rather broadly defined and includes pyrethroids containing descyano-3-phenoxybenzyl or other alcohols. Many of the older non-phenoxybenzyl Type 1 compounds (e.g., bioallethrin, tetramethrin) are unstable in the environment and this characteristic prevented their use in row crops, but they found wide application for use indoors. Introduction of the phenoxybenzyl (e.g., permethrin) or certain halogenated alcohols (e.g., tefluthrin) improved chemical stability and allowed the use of pyrethroids in the field. A more recently developed compound in this group is metofluthrin, specifically designed for biting insect control, and it possesses a strong spatial repellent property and low mammalian toxicity.
The Type 2 pyrethroids are more narrowly defined in terms of their chemical structure. They specifically contain an a-cyano-3-phenoxybenzyl alcohol (-CN, Fig. 1), which increases insecticidal activity about 10-fold, compared to the cognate non-cyano analogs. Moreover, some commercially important Type 2 pyrethroids have altered the acid portion of the molecule to include a phenyl ring (e.g., fenvalerate and fluvalinate). The stereoisomerism of pyrethroids is important for their toxic action, but a detailed discussion of this topic is beyond the scope of this course on IPM. Recent developments in the area of pyrethroid chemistry include the introduction of new active ingredients having isomeric compositions enriched in toxic isomers, designated by Greek letters (e.g., beta cypermethrin). For a detailed discussion of this isomeric enrichment, the reader is directed to the book chapter titled “Pyrethroids” by Khambay and Jewess in Insect Control, Biological and Synthetic Agents (2010).
The signs of intoxication by pyrethroids develop rapidly and there exist different poisoning syndromes for the two types of compounds. Typical signs of intoxication by Type 1 pyrethroids include hyperexcitability and convulsions in insects and a whole body tremor or T syndrome in mammals. In insects, the Type 2 pyrethroids cause predominantly ataxia and incoordination, while in mammals they produce the CS syndrome consisting of choreoathetosis (sinuous writhing) and salivation. In insects, the effects of pyrethroids (especially Type 1) can develop within 1-2 minutes after treatment and can result in knockdown, which is a loss of normal posture and locomotion. Human dermal exposure to either type of pyrethroid can cause paresthesia, a tingling or burning sensation of the skin, but this effect is more intense for Type 2 compounds.
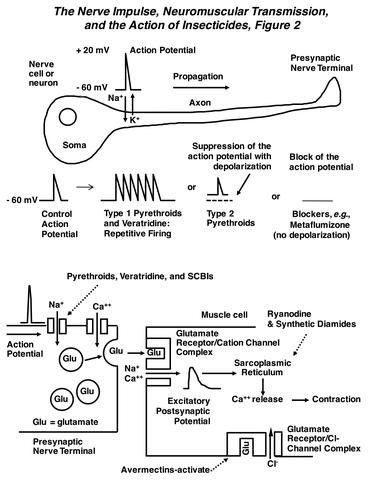
Pyrethroid intoxication results from their potent effects on nerve impulse generation within both the central and peripheral nervous systems. Under normal conditions, neurons possess a transmembrane voltage of about -60 mV on the inside. The nerve impulse or action potential consists of a transient depolarization (positive wave) whose upstroke is driven by an influx of Na+ ions, followed by a downstroke (repolarization) from the efflux of K+ ions. These ion fluxes occur due to the opening and closing of specific ion channel proteins embedded within the nerve membrane. The action potential is propagated down the axon until it reaches the nerve terminal, where it stimulates the release of chemical transmitters. Type 1 compounds induce multiple spike discharges in peripheral sensory and motor nerves, as well as interneurons within the central nervous system (CNS). In contrast, Type 2 pyrethroids depolarize the axon membrane potential, which reduces the amplitude of the action potential and eventually leads to a loss of electrical excitability. All these effects occur because pyrethroids prolong the current flowing through sodium channels by slowing or preventing the shutting (inactivation) of the channels. The somewhat different actions observed for Type 1 and Type 2 compounds are due to differences in the degree of physiological effect: the duration of modified sodium currents by Type 1 compounds lasts tens or hundreds of milliseconds, while those of Type 2 compounds last for minutes or longer. These effects on the sodium current also cause a profound increase in the release of neurotransmitters from presynaptic nerve terminals. The insect neuromuscular synapse is an especially important target for the pyrethroids, as well as other insecticides, and uses glutamate as a neurotransmitter (The Nerve Impulse, Neuromuscular Transmission and the Action of Insecticides, Figure 2). A new structure-activity model of pyrethroids has also appeared, indicating that the miticidal pyrethroid binding pocket is a bit longer than that of insects, which would explain the differential toxicity of pyrethroids active against mites. These findings have important implications for control of varroa mites in bee hives, where the standard compound is fluvalinate. Other experimental results have implicated a role for voltage-sensitive calcium and chloride channels in pyrethroid action in mammals, but detailed discussion of these effects is beyond the scope of this chapter.
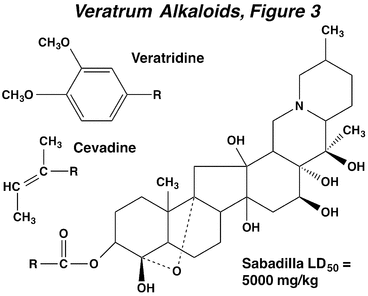
When used in organic farming or gardening, the veratrum alkaloids are usually applied as an extract from the seeds of sabadilla, (Schoenocaulon spp., Liliaceae). The insecticidal activity of sabadilla comes from the alkaloid fraction, which constitutes 3-6% of the extract. The two most important compounds are the lipophilic alkaloids veratridine and cevadine, with veratridine having greater insecticidal potency. The ether linkage (dashed line) in these compounds is critical for activity. Sabadilla breaks down rapidly in sunlight.
Sabadilla extract causes somewhat similar signs of intoxication in insects and mammals. Major effects include muscle rigor in mammals and paralysis in insects. In addition, sabadilla strongly irritates mucous membranes in mammals and causes violent sneezing. Sabadilla extract is much less toxic to mammals than most other insecticides and therefore is relatively safe to use.
The mode of action of the veratrum alkaloids is similar to that of the pyrethroids. When applied to nerve, veratridine causes an increase in the duration of the action potential, repetitive firing, and a depolarization of the nerve membrane potential (The Nerve Impulse, Neuromuscular Transmission and the Action of Insecticides, Figure 2). Depolarization of neurons by veratridine requires the presence of extracellular sodium ions and can stimulate the release of various neurotransmitters. Veratridine prolongs the open state of sodium channels by increasing the probability of channel opening, and then delaying channel inactivation. Interestingly, the simultaneous actions of veratridine and pyrethroids on the sodium channel are synergistic.
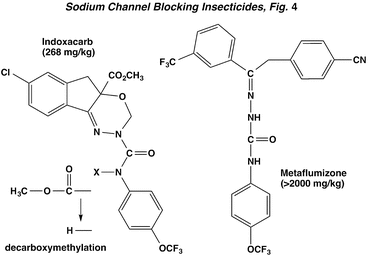
These sodium channel blocking insecticides (SCBI) include the oxadiazine, indoxacarb, and the semicarbazone, metaflumizone. Indoxacarb and metaflumizone tend to have a slower action than pyrethroids, and may cause progressive mortality over several days, depending on the species and route of exposure. Indoxacarb requires metabolic decarboxymethylation in order to generate the actual toxicant, colloquially designated as DCJW. Signs of intoxication develop slowly after topical application to lepidopteran larvae, and include ataxia, paralysis, and curling, with tremors and convulsions occurring when the insects are disturbed. A similar quiescent phase with stimulated convulsions is observed in American cockroaches.
As for neurophysiological effects, the CNS shows depressed nerve activity in prostrate insects, unlike the hyperexcitation caused by pyrethroids. Nerve discharge blockage is eventually global, with stretch receptor preparations of lepidopteran larvae especially sensitive. More detailed electrophysiological studies showed that SCBIs stabilize an inactivated (non-conducting) state of the voltage-sensitive sodium channel and thereby prevent neuronal activation following an electrical stimulus (The Nerve Impulse, Neuromuscular Transmission and the Action of Insecticides, Figure 2). Lack of electrical excitability in the nerves is responsible for paralysis. The SCBIs are insensitive to the target site resistance (kdr) that evolved from the use of pyrethroids.
Insecticides Targeting Calcium Channels
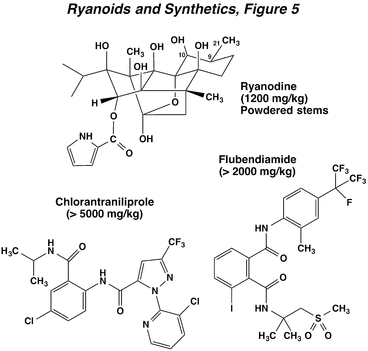
The water-soluble plant extract ryania has been used as an insecticide for over 50 years and consists of the powdered stem of the tropical shrub Ryania speciosa. The extract contains several structurally related ryanoids, including: ryanodine, 10-(O-methyl)-ryanodine, and 9, 21-dehydroryanodine. The most toxic and abundant compounds in the extract are ryanodine and 9, 21-dehydroryanodine, and thus, they account for virtually all of the insecticidal activity. The extract has a low acute toxicity to mammals. New compounds mimicking the action of ryanodine are the anthranilic and phthalic acid diamides, chlorantraniliprole and flubendiamide, respectively. The latter compound is unique in having an iodine ring substituent. These compounds are not chemical analogs of ryanodine, but have similar physiological effects, and even less mammalian toxicity.
Ryanodine and synthetic diamides induce cessation of feeding, lethargy, paralysis and death. These effects occur from induction of a sustained contracture of skeletal muscle without depolarizing the muscle membrane. Under normal conditions, muscle contraction is initiated by the following sequence of events (The Nerve Impulse, Neuromuscular Transmission and the Action of Insecticides, Figure 2). First, an action potential in the motor nerve is conducted into the nerve terminal and this depolarization activates calcium channels and stimulates an influx of Ca++ ions. These ions promote the release of glutamate. The glutamate diffuses across the synaptic cleft, and binds to a receptor-operated ion channel that allows the influx of sodium and calcium ions. This influx induces a depolarization in the muscle membrane that propagates into the muscle fiber via the transverse tubule system to the sarcoplasmic reticulum. The sarcoplasmic reticulum is a calcium storage organelle that when depolarized, releases calcium ions onto the protein filaments responsible for muscle contraction. A number of studies have confirmed that ryanodine, chlorantraniliprole, and flubendiamide can activate the calcium release channel in the sarcoplasmic reticulum. The prolonged opening of this calcium channel floods the muscle fibers with calcium, inducing the sustained contraction of skeletal muscle and paralysis observed in poisoning. In addition, this calcium release channel is also present in some neurons, and responds similarly to these compounds. Because they are not structural analogs of ryanodine, the phthalic and anthranilic diamides work at a different site on the calcium release channel, and do not displace ryanodine from its binding site.
Acetylcholine Mimics
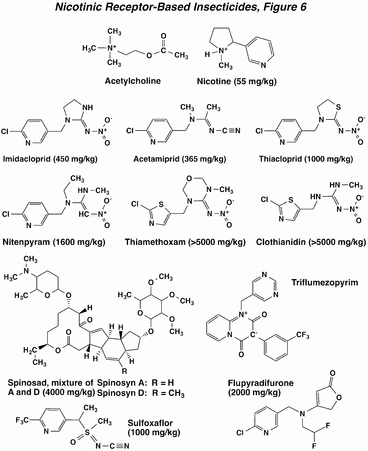
The tobacco alkaloid nicotine has been used as an insecticide since the middle of the 18th century. This compound is miscible with water and is often formulated as the sulfate salt. Nicotine has excellent contact activity, due to its ability to penetrate the integument of insects. This property increases the hazards of handling nicotine, as its contact toxicity to mammals is also appreciable. A significant group of compounds called the neonicotinoids was developed around the structure of the nitroguanidine, imidacloprid. This compound generally works best as a stomach poison, and has plant systemic activity as well, especially against aphids. It is much less toxic to mammals than nicotine. The compound thiamethoxam is apparently converted, in vivo, to clothianidin, which is the actual toxicant. A group of related natural product insecticides are the spinosyns, macrocyclic lactones isolated as fermentation products. Although they differ significantly in structure from the neonicotinoids, these compounds also affect the nicotinic acetylcholine receptor, as given below. Recently introduced compounds that differ in the details of their mode of action on nicotinic receptors are flupyradifurone, sulfoxaflor, and triflumezopyrim, where the latter compound has an unusual internal electronic structure.
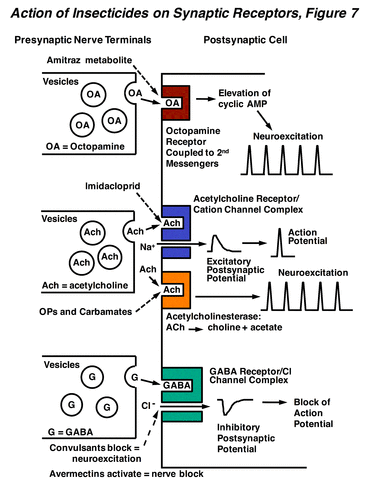
Nicotine and imidacloprid mimic the action of acetylcholine, which is a major excitatory neurotransmitter in the insect CNS. Under normal physiological conditions, after acetylcholine is released by the presynaptic cell, it binds to the postsynaptic nicotinic acetylcholine receptor and activates an intrinsic cation channel. Channel opening results in a depolarization of the postsynaptic cell due primarily to an influx of sodium ions. The synaptic action of acetylcholine is terminated by the enzyme acetylcholinesterase, which rapidly hydrolyzes the ester linkage in acetylcholine. Nicotine, imidacloprid, and related neonicotinoids, also activate the nicotinic acetylcholine receptor, but do so persistently, since they are insensitive to the action of acetylcholinesterase. This persistent activation leads to an overstimulation of cholinergic synapses, and results in hyperexcitation, convulsions, paralysis, and death of the insect. The remaining compounds differ in the details of their actions on the nicotinic acetylcholine receptor, but overall have similar effects.
Acetylcholinesterase Inhibitors
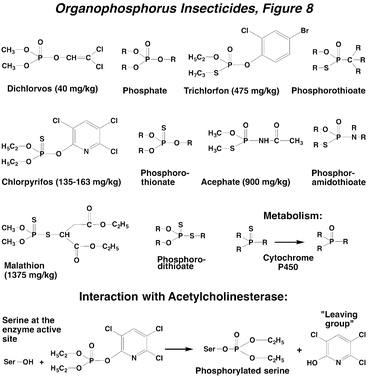
The organophosphorus insecticides (OPs) are a very important group of compounds that vary tremendously in chemical structure and chemical properties. These compounds can be miscible with water, but more typically are miscible in organic solvents. OPs can be classified into several groups depending on the atoms that are directly attached to the central phosphorus. Thus, the majority of important OPs exist in a multitude of chemical configurations (see http://www.alanwood.net/pesticides/class_insecticides.html) and only a few principal compounds are shown. An important bioactivation step occurs for OPs containing a sulfur atom attached to the phosphorus by a double bond. For these compounds, oxidative desulfuration to the oxon form occurs via cytochrome P450 monooxygenases, which are enzymes that breakdown a wide variety of xenobiotics. However, in this case the oxidized metabolite possesses greater chemical reactivity and toxicity; compounds requiring metabolic bioactivation are known as proinsecticides. The acute toxicity of the OPs varies substantially, but historically many of them had high mammalian toxicity, although the most toxic ones have been removed from the market or their uses are restricted.
The primary target site for the OPs is the enzyme acetylcholinesterase. These compounds react with a serine hydroxyl group in the enzyme active site, producing a phosphorylated hydroxyl group and yielding an hydroxylated “leaving group” (Organophosphorus Insecticides, Figure 8). This process inactivates the enzyme and blocks the degradation of the neurotransmitter acetylcholine (Action of Insecticides on Synaptic Receptors, Figure 7). The synaptic concentrations of acetylcholine then build up and hyperexcitation of the CNS occurs, composed of long discharges of action potentials arising from a single stimulus. The signs of intoxication include restlessness, hyperexcitability, tremors, convulsions, and paralysis. In mammals, there is also significant involvement of peripheral cholinergic systems in OP poisoning, resulting in SLUDGE (Salivation, Lachrymation, Urination, Defecation, Gastrointestinal upset, Emesis). In insects, the effects of OPs are confined to the CNS, where virtually all of the cholinergic synapses are located. Because they often require bioactivation and must penetrate into the CNS, the OPs do not have a rapid action like that of the pyrethroids. The phosphorylation of acetylcholinesterase by OPs is persistent, and reactivation of the enzyme by water hydrolysis of the serine-phosphate bond can take many hours or even days.
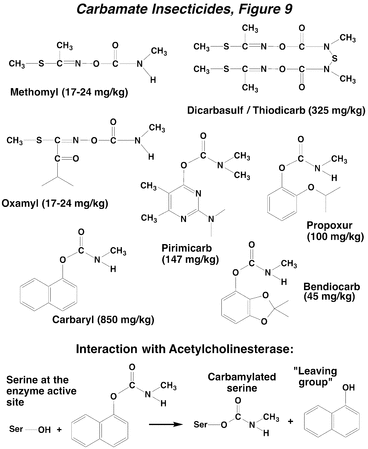
Carbamate insecticides are esters of carbamic acid, having either an aliphatic oxime (C=N-O), such as methomyl or an aryl substituent, such as propoxur. Both types of compounds are soluble in organic solvents. The carbamates are often highly toxic to mammals, and must be handled carefully. Among insects, they can be particularly toxic to beneficial hymenoptera such as honeybees. A clever trick employed with carbamates was to utilize an N-S-N linkage at the carbamate nitrogen (e.g., thiodicarb), which degrades, in vivo, to yield the active parent carbamate. This mechanism reduces mammalian toxicity substantially with little effect upon insecticidal efficacy. The mode of action of the carbamates is similar to that of the OPs. In this case, the reaction yields a carbamylation of the serine hydroxyl group (Carbamates, Figure 9), and a hydroxylated leaving group is again generated. However, the carbamylated form of acetylcholinesterase is not as stable as phosphorylation, and has a half-life of about 40 min. The CNS is the site of action of carbamates, the overall effect is central nervous hyperexcitation (Action of Insecticides on Synaptic Receptors, Figure 7), and the signs of intoxication and effects on synaptic function are also similar to those of the OPs (SLUDGE).
Octopamine Mimics
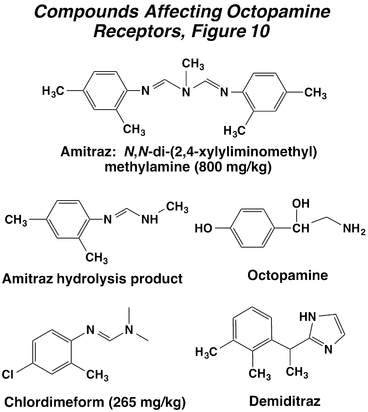
Several amidine compounds have been used for their insecticidal and miticidal properties, as well as an animal treatment for ticks. Chlordimeform was a significant compound in this series, but potential problems with carcinogenicity removed it from the market. The major insecticidal compound still used today is amitraz, which is one of the few compounds effective against varroa mites in bee hives. This material undergoes metabolic hydrolysis to an active metabolite, so it is a proinsecticide. Amitraz is only sparingly soluble in water, but is soluble in organic solvents. Its acute toxicity to mammals is moderate. A new molecule in this group is demiditraz, but it is not widely used commercially.
These compounds affect target species by mimicking the action of the neurotransmitter octopamine, which regulates nerve firing and behavioral arousal levels within the CNS and also has actions on peripheral tissues, especially visceral muscle. Octopamine binds to a receptor that elevates levels of the second messenger cyclic AMP, and this messenger mediates cellular processes that typically give rise to neuronal excitation (Action of Insecticides on Synaptic Receptors, Figure 7). Amidines cause an overstimulation of octopaminergic synapses in insects, resulting in tremors, convulsions, and continuous flight behavior in adult insects. Moreover, these compounds have the ability to cause a true anorexia in insects and also suppress reproduction.
Compounds Affecting Chloride Channels
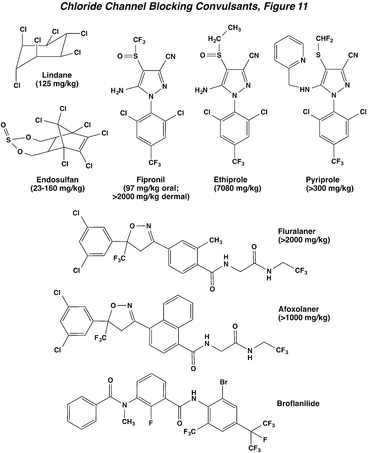
The channel blocking convulsants represent one of the oldest groups of commercial insecticides. Historically, these compounds include the environmentally stable, lipophilic polychlorocycloalkanes, such as heptachlor, dieldrin, and endrin. Only the more biodegradable materials lindane and endosulfan are still applied, but their uses are being phased out. High mammalian toxicity, and especially high dermal toxicity, were hallmarks of the polychlorocycloalkanes. Chemical development efforts resulted in the commercialization of fipronil in the early 1990s, and then later ethiprole and pyriprole, etc., newer arylheterocycles (fiproles) with a similar mode of action, but with lower mammalian toxicity. More recent chemistry has identified active isoxazolines (e.g., fluralaner, afoxolaner, and fluxametamide) and meta-diamides (e.g., broflanilide) having more complicated structures than the older compounds. The isoxazolines were initially introduced as veterinary parsiticides, while the meta-diamides have a broad activity profile, being toxic to a number of disease vectors and agricultural pests. These two novel classes of chemistry show a marked increase in selectivity, as reflected in the much greater rat oral LD50 values than previously observed for members of this group. They also circumvent target site resistance (rdl) that developed to older compounds.
In both insects and mammals, chloride channel-blocking insecticides cause hyperexcitability and convulsions. These effects occur via poisoning of the CNS through antagonism of the inhibitory neurotransmitter g-aminobutyric acid (GABA). Normally, when GABA is released from the presynaptic nerve terminal, it binds to a postsynaptic receptor protein containing an intrinsic chloride ion channel (Action of Insecticides on Synaptic Receptors, Figure 7). When GABA binds to the receptor, the channel is opened and Cl- ions flow into the postsynaptic neuron. This chloride permeability can significantly hyperpolarize (make more negative) the membrane potential and has a dampening effect on nerve impulse firing by reducing membrane resistance. A large number of studies have demonstrated that these insecticides bind to the chloride channel and block its activation by GABA, and this absence of synaptic inhibition leads to hyperexcitation of the CNS. Newer data suggests an additional blocking action of fiproles on glutamate-gated chloride channels, which are also found on insect nerve and muscle (The Nerve Impulse, Neuromuscular Transmission, and the Action of Insecticides, Figure 2), and may contribute to their greater insect selectivity, since these receptors are absent in mammals.
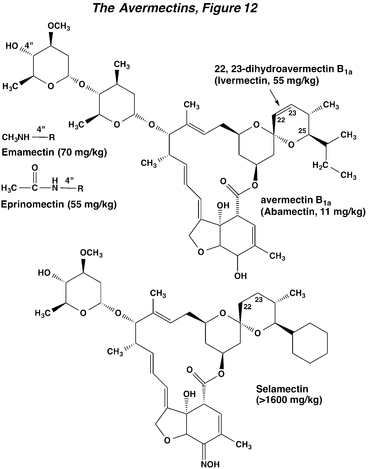
The avermectins are a group of closely related macrocyclic lactones isolated from the fungus, Streptomyces avermitilis. The basic structural motif of the avermectins is evident in the natural product avermectin B1a, which is the principal constituent of the insecticide/miticide abamectin. Chemical modification of avermectin B1a has yielded a number of semi-synthetic materials. One of the most important is the compound emamectin, which has high insecticidal activity against caterpillars. Another is ivermectin, a major compound for controlling worm parasites in veterinary medicine. The avermectins are insoluble in water. Both abamectin and emamectin have fairly high mammalian toxicity, but their translaminar movement into treated leaves, high oral activity against insect pests, and rapid breakdown in sunlight are all favorable properties from an IPM standpoint. Related materials are the milbemycins. Newer materials, such as selamectin (used to control fleas, ticks, and heartworms), have reduced mammalian toxicity.
Avermectin intoxication in mammals begins with hyperexcitability, tremors, and incoordination, and later develops into ataxia and coma-like sedation. In insects and nematodes poisoned by avermectins, ataxia and paralysis are the major signs of intoxication, with little or no hyperexcitation. The avermectins block electrical activity in vertebrate and invertebrate nerve and muscle by increasing the membrane conductance to chloride ions. The effect is similar to that of GABA, but is essentially irreversible (Action of Insecticides on Synaptic Receptors, Figure 7). In tissues containing GABA receptors, the avermectin-dependent conductance increase is often accompanied by a loss of sensitivity to exogenously applied GABA and this GABA blocking action is probably responsible for the transient tremor observed in mammals. Especially important are the glutamate-gated chloride channels of insect and nematode skeletal muscle, which mediate avermectin-induced muscle paralysis in these organisms (The Nerve Impulse, Neuromuscular Transmission, and the Action of Insecticides, Figure 2). Mutation of this receptor can cause resistance to avermectins, indicating its primary role in toxicity to invertebrates.
Disruption of Energy Metabolism
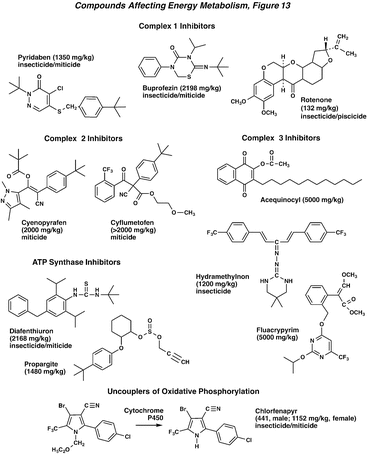
Compounds that disrupt energy metabolism have been identified from both natural and synthetic sources, and in this figure are organized by site of action on the mitochondria. The prototypical member of the Complex 1 inhibitors is rotenone, which is derived from Cube or Derris root. Synthetic compounds in this group include a number of nitrogen-containing heterocycles, such as the miticides fenazaquin and pyridaben. Complex 2 inhibitors are exemplified by cyenopyrafen and cyflumetofen miticides. The Complex 3 inhibitors are structurally diverse, and include acequinocyl, hydramethylnon (ant and cockroach baits), and fluacrypyrim. The ATP synthase inhibitors are represented by the proinecticide/miticide diafenthiuron and the miticide propargite. A commonly used uncoupler of oxidative phosphorylation is chlorfenapyr, a proinsecticide that is cleaved to the active pyrrole. All of these materials have low solubility in water, and they are of low to moderate toxicity to mammals. It is interesting to note that the highest acute toxicity is observed for the natural product rotenone. Due to concerns about possible human toxicity, insecticidal uses of rotenone were phased out in the U.S.A. This compound is also highly toxic to fish, and is still used as a pesticide.
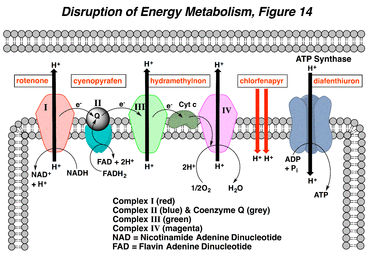
Disruption of energy metabolism occurs in the mitochondria and usually takes the form of either an inhibition of the electron transport system (ETS), blockage of ATP synthesis, or an uncoupling of the transport system from ATP production. The ETS (Complexes I-IV as shown left to right) are macromolecular complexes that use high energy electrons to pump hydrogens into the intermembrane space and to store this energy as a proton gradient, which is harnessed to synthesize ATP. Oxygen is the terminal acceptor of these protons, forming water as a by-product. Inhibition of the ETS blocks indirectly the production of ATP and causes a decrease in oxygen consumption by the mitochondria. Rotenone, fenazaquin, and pyridaben are inhibitors at Complex I in the ETS, while the other inhibitors affect Complexes II (e.g., cyenopyrafen) and III (e.g., hydramethylnon), as indicated by red text boxes (Figure 14). Blockage at all these sites can reduce energy production and induce whole animal toxicity. Inhibition of ATP-synthase directly reduces ATP production (e.g., diafenthiuron), but without affecting the ETS. For an uncoupling action, the transport system functions normally, but the production of ATP is “uncoupled” from the electron transport process due to a dissipation of the proton gradient across the inner mitochondrial membrane (e.g., chlorfenapyr). Protons are shuttled across the membrane, oxygen consumption increases, but little ATP is produced (red arrows, Figure 14). The disruption of energy metabolism and the subsequent loss of ATP results in a slowly developing toxicity, and the effects of all these compounds include inactivity, paralysis, and death.
Insect Growth Regulators
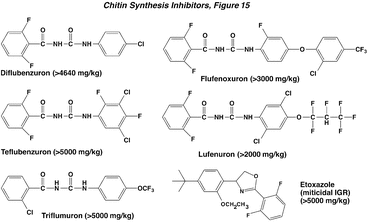
The original compounds having this mode of action were the benzoylphenylureas and possess a number of ring halogen substituents. Diflubenzuron is the prototypical compound in this series, and a number of subsequent compounds were also developed as insecticides. The water solubility of these materials is typically extremely low (< 1ppm), as is their mammalian toxicity. Newer miticidal chitin synthesis inhibitors have deviated from this structural motif (e.g., etoxazole).
Insects exposed to these compounds are unable to form normal cuticle because the ability to synthesize/assemble chitin is lost. About 50% of the cuticle is comprised of chitin, which is a polysaccharide of N-acetylglucosamine units. This polymerization is blocked by the benzoylphenylureas, and may occur through inhibition of a membrane transport step involving UDP-N-acetylglucosamine. These compounds are not inhibitors of chitin synthetase. In the absence of sufficient chitin, the cuticle becomes thin and brittle, and is unable to support the insect or to withstand the rigors of molting. Accordingly, the benzoylphenylureas are especially effective when applied just before a molt. They tend to have little effect during the intermolt interval, and there is a time sensitive period when they are most effective. These compounds will work against any other organisms with chitinous exoskeleton, such as freshwater crayfish. Illustration of the cuticular effects of triflumeron and etoxazole can be seen in the paper by Nauen and Smagghe (2006).
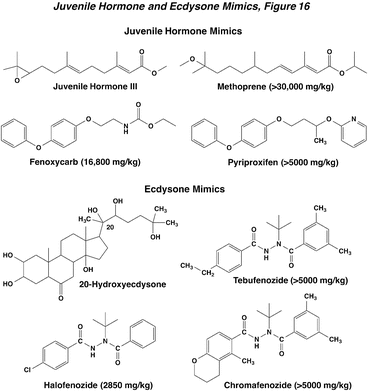
Methoprene is the pioneer juvenile hormone analog and the most prominent. It is an effective inhibitor of emergence of adult mosquitoes at low concentrations (10 ppb). The material has a short half life in the environment, and it can also be used indoors for flea control. Second generation compounds include fenoxycarb and pyriproxifen. All these compounds have low mammalian toxicity. They mimic the action of applied juvenile hormone, but are not subject to the same biochemical regulatory mechanisms. They interfere with the molting process, often resulting in pupal/adult intermediates, or similar disruptive effects on development and molting. The failure to undergo pupal or adult molt can producing supernumerary larvae or nymphs. For example, a 6th instar larva is produced when normally there are only five. They can also be ovicidal, and can induce production of queen imaginal disks in worker honey bee larvae. There are prominent effects in adult German cockroach, including over-melanization, crinkled wings, and failure of the reproductive organs. Here again, treatment timing is critical: it must be present during a period late in the life cycle to be effective.
The ecdysteroid mimics belong to a chemical class called diacylhydrazines, with the original commercial compound named tebufenozide. This compound was followed by other analogs, such as halofenozide, and chromafenozide. All have a signature tert-butyl substituent on the diacylhydrazine moiety. Tebufenozide and chromafenozide are highly selective on caterpillars, while halofenozide is also active against coleoptera. They all have very low mammalian toxicity. These are non-steroidal compounds, but mimic the action of 20-hydroxyecdysone (the active metabolite of ecdysone). When exposed via the diet, caterpillar feeding stops after 24 hr, and there is premature slippage of the head capsule. Head capsule slippage shuts down ecdysone production, molting, and larval growth. There is a good correlation between the potency of analogs for affecting head capsule slippage and ability to displace the binding of labeled hormone from insect cells. The morphological and physiological effects of ecdysteroid mimics can be viewed in more detail in the original paper by Wing et al. (1988).
Acknowledgements
I would like to thank Dr. William Hutchison of the Department of Entomology, University of Minnesota, for his kind acceptance of this revised chapter.
References
Biochemical Sites of Insecticide Action and Resistance. I Ishaaya, Ed., pp. 343, Springer-Verlag, Berlin Heidelberg, Germany (2001).
JR Bloomquist, Ion Channels as Targets for Insecticides. Ann. Rev. Entomol. 41, 163-190 (1996).
JM Clark, JG Scott, F Campos, and JR Bloomquist, Resistance to Avermectins: Extent, Mechanisms, and Management Implications. Ann. Rev. Entomol. 40, 1-30 (1995).
BPS Khambay and PJ Jewess, “Pyrethroids” In: Insect Control. Biological and Synthetic Agents. LI Gilbert and SS Gill, Eds., pp. 470, Academic Press, London, United Kingdom (2010).
R Nauen and G Smagghe, Mode of action of etoxazole. Pest Manag Sci 62:379–382 (2006).
DM Soderlund, JM Clark, LP Sheets, LS Mullin, Vincent J. Piccirillo, Dana Sargent, James T. Stevens, Myra L. Weiner. Mechanisms of pyrethroid neurotoxicity: implications for cumulative risk assessment. Toxicology 171, 3-59 (2002).
GW Ware and DM Whitacre, The Pesticide Book, 6th Edition. pp. 488, MeisterPro, Willoughby, Ohio (2004).
KD Wing, RA Slawecki, GR Carlson, RH-5849, a non-steroidal ecdysone agonist, effects on larval Lepidoptera. Science 241:470-472 (1988)
S Yu, The Toxicology and Biochemistry of Insecticides, Second Edition, pp. 380, CRC, Boca Raton (2014).