Extracted from The Pesticide Book, 6th ed*. (2004), Published by MeisterPro Information Resources, A division of Meister Media Worldwide, Willoughby, Ohio
George W. Ware
Professor Emeritus
Department of Entomology
University of Arizona
David M. Whitacre
V.P. Development (Retired)
Novartis, Syngenta Crop Protection
Structural formulae shown in this chapter are reproduced with permission from Alan Wood's Compendium of Pesticide Common Names
Herbicides, chemicals designed specifically to kill weeds, account for nearly 60% of all pesticide sales in the U.S. U.S. farmers spent an estimated $5.63 billion in 1998 for herbicides, with about $1.1 billion more spent in application costs. Herbicide use declined in 1999 to only a little more than $5.0 billion (Donaldson et. al., 2002).
Weeds are objectionable to humans primarily because they reduce the quality and quantity of agricultural production, and produce allergens or contact dermatitis that affect public health. Approximately 10% of all plant species are weeds, or a total of some 30,000 weed species. Of these, 1,800 cause serious economic losses in crop production, and about 300 species plague cultivated crops throughout the world. The U.S. has become home to 70% of the world’s worst weeds.
Herbicides, or chemical weed killers, have largely replaced mechanical methods of weed control in countries where intensive and highly mechanized agriculture is practiced. Herbicides provide a more effective and economical means of weed control than cultivation, hoeing, and hand pulling. Together with fertilizers, other pesticides, and improved plant varieties, they have made an important contribution to the increased yields we now have and serve to combat rising costs and shortages of agricultural labor. The heavy use of herbicides, although growing in many developing countries, is largely confined to North America, Western Europe, Latin America, Japan, and Australia. Without the use of herbicides, it would have been impossible to mechanize fully the production of cotton, sugar beets, grains, potatoes, and corn.
Herbicides are also used extensively away from the farm, in areas such as industrial sites, roadsides, ditch banks, irrigation canals, fence lines, recreational areas, lawns, railroad embankments, and power line rights-of-way. Herbicides remove undesirable plants that might cause damage, present fire hazards, or impede work crews. They also reduce costs of labor for mowing.
Herbicides are classed as selective when they are used to kill weeds without harming the crop and as nonselective when the purpose is to kill all vegetation. Both selective and nonselective materials can be applied to weed foliage or to soil containing weed seeds and seedlings, depending on the mode of action, and intended use pattern. The term true selectivity refers to the capacity of an herbicide, when applied at the proper dosage and time, to be active only against certain species of plants but not against others. But selectivity can also be achieved by placement, as when a nonselective herbicide is applied such that it reaches the weeds but not the crop.
Herbicide classification would be a simple if only the selective and nonselective categories existed. However, there are multiple classification schemes that may be based on selectivity, contact versus translocation, timing, area covered, and chemical classification.
Another scheme classifies herbicides into either contact or translocated categories. Contact herbicides kill the plant parts onto whose surfaces the chemical is applied and are most effective against annuals, those weeds that germinate from seeds and grow to maturity each year. Complete coverage is essential in weed control with contact materials. Translocated herbicides are absorbed either by roots or above-ground parts of plants and are circulated within the plant system to distant tissues. Translocated herbicides may be effective against all weed types; however, their greatest advantage is in the control of established perennials, those weeds that continue their growth from year to year. Uniform application is a key requirement in achieving good weed control with translocated herbicides.
Herbicides are also classed according to timing of application, i.e., growth stage of crop or weed development. What constitutes the best timing varies by chemical class of the material and its persistence, the crop and its tolerance to the herbicide, weed species, cultural practices, climate, and soil type and condition. The three primary categories of application timing are preplanting, preemergence, and post emergence.
Preplanting applications for control of annual weeds are made to an area before the crop is planted, within a few days or weeks of planting and often includes a step to incorporate the herbicide in the soil. In recent years such preplant uses have included making fall applications of herbicides to suppress early weed growth the next spring before corn is planted. Preemergence applications are completed prior to emergence of the crop or weeds, depending on definition, after planting. Postemergence applications are made after the crop or weed emerges from the soil.
Herbicide application based on the area covered involves four categories: band, broadcast, spot treatments, and directed spraying. A band application treats a continuous strip, as along or in a crop row. Broadcast application covers the entire area, including the crop. Spot treatments are confined to small areas of weeds. Directed sprays are applied to crop stems, selected weeds or to the soil to avoid contact with crop foliage.
Over-the-top-application occurs when herbicides are applied over-the-top on the crop and weeds shortly after germination. The crops in these instances are naturally tolerant to the specific herbicide or have been genetically engineered to be tolerant. Examples of crops bioengineered to sustain broad spectrum herbicides include new varieties of cotton, corn, soybeans, canola, rice, sugarbeets, wheat and turf.
Inorganic Herbicides
The first chemicals used in weed control were inorganic compounds. Brine and a mixture of salt and ashes were both used by the Romans to sterilize the land as early as biblical time. In 1896, copper sulfate was used selectively to kill weeds in grain fields. From about 1906 until 1960, sodium arsenite solutions were the standard herbicide of commerce. Arsenic trioxide has been used at rates of 400 to 800 lbs/acre for soil sterilization, whereas 1 or 2 lbs of currently used, selected organic herbicides would easily produce the same result. The trivalent arsenicals are nonspecific inhibitors of those enzymes containing sulfhydryl groups, and also uncouple oxidative phosphorylation.
Ammonium sulfamate (NH4SO3NH2), another salt, was introduced in 1942 for brush control. Other salts have been used over the years. The borate herbicides (sodium tetraborate, sodium borate, etc.) were used heavily for soil sterilization. A nonselective herbicide used extensively for the last 50 years is sodium chlorate (NaClO3). It acts as a soil sterilant at rates of 200 lbs/acre, but it can be used as a foliar spray at 5 lbs/acre as a cotton defoliant. Several of the inorganics are still useful in weed and brush control, but are rapidly being replaced by organic materials. EPA has placed heavy restrictions on some inorganics because of their persistence in soils. Advice to the reader: inorganics are not wise choices for use around the home except by experts intending to remove all vegetation from an area.
Organic Herbicides
The categorization of organics is rather complex, comprising some 31 classes, if the old, now illegal, category of petroleum oils is included. The classification followed in this chapter is generally based on the Herbicide Classification of the Weed Science Society of America (Weed Technology 11:384-393, 1997).
Chloroacetamides (formerly Acetanilides)
The chloroacetamides and amides are both meristematic growth inhibitors. They differ in that the amides are not translocated.
The chloroacetamides are used as preemergence herbicides, some preplant soil incorporated and others surface applied. Of these, alachlor was one of the first to gain wide acceptance. In 1999, it was the 17th most used pesticide, in the nation with an estimated 7-10 million pounds being used in agriculture, mainly on corn, cotton and soybeans.
Metolachlor (Dual Magnum®) is soil-incorporated as a preplant and preemergence herbicide for annual grass and some broadleaf weeds in corn, soybeans and peanuts. Metolachlor and a more recent resolved isomer form (s-metolachlor) together accounted for a total use of 42-50 million pounds in 1999 (Donaldson et. al., 2002) Acetochlor (Harness®, Surpass®, TopNotch®), not illustrated, was first registered for crop use in 1994. In 1999, more than 30 million pounds of acetochlor was used in US agriculture, mostly on corn. Butachlor is used only on rice, but not in the U.S., to control grass weeds and is applied either preplant incorporated or preemergence to seeded or transplant rice. Propachlor is used on corn, cotton, sorghum and soybeans to control mostly grass weeds. It is applied to the soil surface prior to weed and crop emergence. One new member of this class has surfaced recently. It is thenylchlor (Kasamets®) which is slated for use on rice in South Korea.
METOLACHLOR (Dual MAGNUM®)
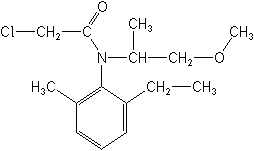
The mode of action for this class is predominantly inhibition of very long chain fatty acids.
Amides
The amide herbicides have diverse biological properties. Amides are sometimes grouped with the chloroacetamides. Dimethenamid (Frontier®), first registered in 1993, controls the major grass weeds, broadleafs and yellow nutsedge in corn, soybeans, peanuts, dry beans and sorghum as a selective preemergence herbicide. A resolved form, in which the product was purified to yield the active s isomer, was developed and registered in 1999. This product is named dimethenamid-P (Outlook®).
Propanil (Stampede®) has been used extensively on rice fields as a selective postemergence control for a broad spectrum of weeds. Napropamide is used in the control of grass and broadleaf weeds in vineyards and orchards, and in direct-seeded tomatoes, strawberries, ornamentals, and tobacco.
PROPANIL (Stampede®)
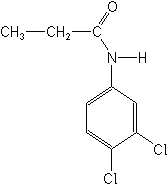
Propanil acts primarily in the leaves and is a strong inhibitor of the Hill reaction. This is a light-initiated reaction that splits water (photolysis), resulting in the production of free oxygen (O2) by plants. Chlorophyll, the green pigment of plants, is an essential ingredient in the reaction, since it catalyzes the production of oxygen from water and the transfer of the hydrogen to a hydrogen-acceptor.
Naptalam is used as preemergence sprays for controlling germinating seedlings of both grasses and broadleaf weeds. Its mode of action is the blocking of the growth control regulator indoleacetic acid. Pronamide is used as a selective, preemergence and early postemergence herbicide, for both grass and broadleaf weeds in a broad range of crops. It is taken up by roots, and has little foliar activity. Bensulide is one of the better turf herbicides, especially for the control of crabgrass. It could be classified as an organophosphate herbicide, and has higher mammalian toxicity than most herbicides because it is an organophosphate. Bensulide acts by inhibiting cell division in root tips. It is used as a preemergence herbicide in lawns to control certain grasses and broadleaf weeds, but it fails as a foliar spray because it is not translocated
A new member of this class is pethoxamid (Korban®, Successor®), destined for use in Europe for weed control in corn and soybeans. This product, like dimethenamid, is an inhibitor of very long chain fatty acids.
There is no consistent pattern in the mechanism of action of amide herbicides. Some are applied only to the soil and are active through the root system or seeds, while others are applied only to foliage.
Organoarsenicals
The organic arsenicals are no longer widely used. The class includes derivatives of arsinic and arsonic acids. Cacodylic acid (dimethylarsinic acid) and its sodium salt are the only derivatives of arsinic acid. Disodium methanearsonate (DSMA) and monosodium methanearsonate (MSMA) are salts of arsonic acid.
MSMA
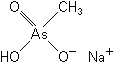
The organic arsenicals are much less toxic to mammals than the inorganic forms, are crystalline solids, and are relatively soluble in water. The arsonate, or pentavalent arsenic, acts in a way different from the trivalent form of the inorganic arsenicals just described. The arsonates upset plant metabolism and interfere with normal growth by entering into reactions in place of phosphate. They not only substitute for essential phosphate but also are absorbed and translocated in a manner similar to that of phosphates.
The trivalent arsenates are exclusively contact herbicides, whereas the pentavalent arsonates are translocated to underground tubers and rhizomes, making them extremely useful against johnsongrass and nut sedges. They are often applied as spot treatments.
Benzoic Acids
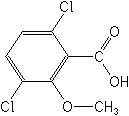
Several derivatives of benzoic acid are employed as herbicides and are applied to the soil against germinating seeds and seedlings. They are classed as growth hormone herbicides, and move both from leaves to the terminal meristems of leaf, shoot and root, and also move in the transpiration stream, permitting them to also be soil applied. Their mode of action is thought to be the interference in protein synthesis with the resulting interference in cell formation. Dicamba has been one of the 25 most used pesticides in the U.S. for nearly two decades. Recent annual usage rates have been 6-8 million pounds. Chlorfenac and Chloramben have been discontinued.
DICAMBA (Banvel®)
Nitriles (formerly Benzonitriles)
Nitriles have a benzene ring, containing the CN or cyanide grouping. These two compounds, dichlobenil and bromoxynil, are contact herbicides which control broadleaf weeds in grass-type crops. They are registered for use on small grains, corn, grain sorghum, and for seedling turf for postemergence control of broadleaf weeds.
DICHLOBENIL (Casoron®)
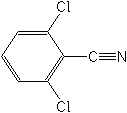
Their mechanisms of action are broad, involving seedling growth inhibition, potato sprout inhibition, and gross disruption of tissues by inhibiting oxidative phosphorylation and preventing the fixation of CO2. These effects, however, do not explain their rapid action. Ioxynil belongs to the nitriles but is not registered in the U.S.
Benzothiadiazoles
The single representative of this group is bentazone. Bentazone kills broadleaf weeds and nutsedge by inhibiting photosynthesis. It differs from other photosynthesis-inhibitors, such as the triazines and phenylureas, in that very limited translocation occurs. Consequently uses are limited to postemergence treatments and require thorough plant coverage to be effective. Bentazon is effective in controlling broadleaf weeds in soybeans, rice, corn, peanuts, beans, and peas.
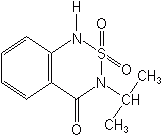
BENTAZONE (Basagran®)
Bipyridyliums
The name bipyridylium suggests the attachment of two pyridyl rings. There are three important herbicides in this group, diquat, paraquat and difenzoquat. All are contact herbicides that damage plant tissues quickly, causing the plants to appear frostbitten because of cell membrane destruction. This rapid wilting and desiccation occur within hours, making these novel herbicides also useful as preharvest desiccants for seed crops, cotton, soybeans, sugarcane, and sunflowers. Diquat is also used in aquatic weed control. None are active in soils.
The action of these herbicides occurs through positive ions, naturally dissociated, which are reduced by photosynthesis to form stable free radicals. These free radicals are then oxidized to reform the original ion and hydrogen peroxide which destroys plant tissue.
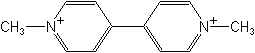
PARAQUAT
Carbamates
The carbamates are esters of carbamic acid, and some representatives are active as herbicides, and some as fungicides or insecticides. Discovered in 1945, the carbamates described here are used primarily as selective preemergence herbicides, but some are also effective in postemergence use.
The first of the herbicidal carbamates was propham (also known as IPC); it was followed by chlorpropham and later by asulam. These herbicides kill plants by stopping cell division and plant tissue growth. Two effects are noted: cessation of protein production and shortening of chromosomes undergoing mitosis (duplication). Carbamates are readily translocated and inhibit meristematic development.
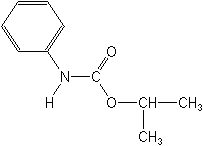
PROPHAM (Turberite®)
Phenyl-carbamates
Similar to the carbamates are the phenyl-carbamates, of which there are but two. Phenmedipham contains two carbamate radicals in its molecule and is used as a postemergence herbicide in sugar beet & sunflower production. Desmedipham (Betanex®) is registered only for sugar beets in the U.S., for a wide range of broadleaf spring weeds.
PHENMEDIPHAM (Betanal®)
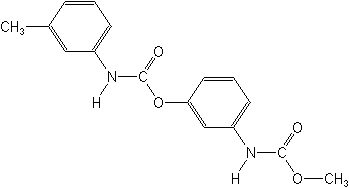
Chlorinated Aliphatic Acids
Two aliphatic acids heavily used as herbicides were TCA and dalapon, used against grasses, particularly quackgrass and Bermudagrass. They both act by precipitation of protein within the cells. Dalapon was widely used around homes to control Bermudagrass. Neither product is marketed in the U.S. today.
Cyclohexanediones
The cyclohexanediones are all selective, postemergence herbicides used for the control of annual and perennial grass weeds in broadleaf crops, e.g., cotton, soybeans, potatoes, sugarbeets, and vegetables. They do not control broadleaf weeds or sedges. Only two materials are registered in the U.S., sethoxydim, and clethodim. Cycloxydim and tralkoxydim are not registered but one new member of the class has recently made an appearance. It is tepraloxydim (Aramo®) which controls various grass weeds in cotton, onions, soybeans and sugarbeets. Although these herbicides do interfere with auxins their primary action is to inhibit lipid biosynthesis.
SETHOXYDIM (Poast®, Vantage®)
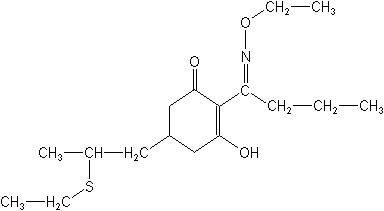
Dinitroanilines
The dinitroanilines are some of the most heavily used in agriculture, and are used almost exclusively as soil-incorporated, preemergence selective herbicides in many field crops. Examples of substituted dinitroanilines are trifluralin, benefin, oryzalin, pendimethalin, and isopropalin. Trifluralin has very low water solubility, which minimizes leaching and movement from the target. The nitroanilines inhibit both root and shoot growth when absorbed by roots, but they have an involved mode of action, which includes inhibiting the development of several enzymes and the uncoupling of oxidative phosphorylation.
TRIFLURALIN (Treflan®)
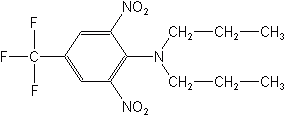
Oryzalin controls annual grasses and broadleaf weeds in rice, cotton, soybeans, nonbearing fruit trees, nut trees, vineyards, and ornamentals. Pendimethalin controls annual grass and certain broadleaf weeds in corn and is preplant incorporated in cotton, soybeans, vegetables, and tobacco. As preemergence herbicides, the dinitroanilines severely inhibit growth of seedling roots, resulting in plant stunting. Prodiamine (Barricade®) is used for turf and ornamentals and Ethalfluralin (Sonalan®), a newer nitroaniline, for cotton and soybeans. It is soil-incorporated selective for annual grasses and broadleaf weeds, and especially useful against nightshade.
Dinitrophenols
The dinitrophenols included DNOC, DINOSEB (Dinitro®, DNBP), and some lesser known materials. These were used for more than 50 years as herbicides, ovicides, fungicides, insecticides, and blossom-thinners. The dinitrophenols act by uncoupling oxidative phosphorylation, not only in plants, but also in warm blooded animals, including man. Consequently, by general agreement with the manufacturers, EPA cancelled all uses of this herbicide class in 1987, because of suspected long term health effects in man.
DINOSEB
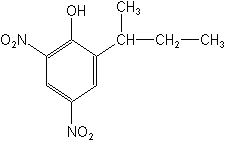
Diphenyl Ethers
The diphenyl ether herbicides are constructed around a molecule made of two phenyl (benzene) rings tied together with oxygen.
The first of this group was nitrofen, introduced in 1963. They are generally used in preemergence or early postemergence weed control. They are primarily contact herbicides and are readily absorbed by leaves and roots. Translocation is limited. They induce chlorosis and necrosis as visible effects of activity. Their mode of action is thought to be inhibition of electron transport and coupled photophosphorylation. In some instances they induce growth responses, resembling the action of auxins and the phenoxy herbicides.
Some of the more successful herbicides are found in this group. They are foliar applied, do not translocate, and are enhanced by high volume sprays containing surfactants to increase coverage. They are used to control broadleaf and grass weeds in soybeans, peanuts, and other legumes, postemergence weed control in wheat, barley, soybeans and sugar beets. They include acifluorfen, lactofen, and oxyfluorfen. Two others are bifenox (Modown®), and fomesafen (Reflex®, Tornado®).
ACIFLUORFEN (Blazer®)
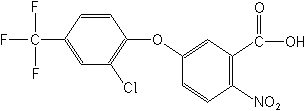
Imidazolinones
The imidazolinones are a relatively new class, first appearing in 1981.The most recent imazamox (Raptor®) was introduced in 1998. They are meristematic inhibitors which inhibit amino acid branched-chain biosynthesis. They are more effective against broadleaf weeds than grasses, and have both foliar and soil activity. Soil persistence ranges from moderate to long.
Imazapyr has been used the longest, as a nonselective, broad-spectrum herbicide with residual activity in noncrop areas and forestry. Imazaquin is a selective pre- and postemergence herbicide which has enjoyed wide use on soybeans. Imazamethabenz-methyl is a selective postemergence herbicide against wild oats, mustards and buckwheat in sunflowers, wheat and barley. Imazethapyr is a preemergence, preplant incorporated and postemergence material for control of annual and perennial grass and broadleaf weeds in soybeans and other legumes.
IMAZAPYR (Arsenal®)
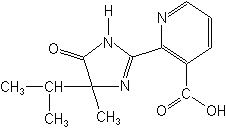
Triazolopyrimidines
This class, closely related to the imidazolinones, were introduced by Dow AgroSciences in 1990. Flumetsulam (Broadstrike®) was the first to be introduced, followed by metosulam (Eclipse®), chloransulam (Firstrate®), and diclosulam (Strongarm®). Flumetsulam is a selective preplant, preemergence and postemergence material for tough broadleaf weeds in corn and soybeans, and sold in the U.S. only as mixtures with other herbicides. Metosulam, similar in selectivity, is not registered in the U.S. Chloransulam and diclosulam are being developed in the U.S. for broadleaf weed control on soybeans and peanuts. New entrants to the class are penoxsulam and florasulam (Primus®); the latter is in development to control broadleaf weeds in corn and cereals and the former is slated for use on rice. Their mode of action is similar to that of the sulfonylureas and imidazolinones.
FLUMETSULAM (Broadstrike®)
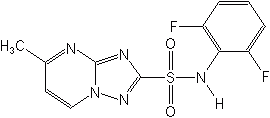
Aryloxphenoxy Propionates (formerly Oxyphenoxy Acid Esters)
This recently introduced class of herbicides is a family of postemergents with high activity against all grasses at economical rates. Consequently, they are applied to most broadleaf crops with little risk of crop injury. At higher rates of application some soil activity is observed. In grasses the compounds are readily translocated from point of uptake to the growing meristem. Weather apparently has little effect on activity. Notably, they are all effective against the arch enemies crabgrass and johnsongrass. Fluazifop-butyl is the most widely used of the group, for postemergence control of both perennial and annual grass weeds, over-the-top in broadleaved crops, especially cotton and soybeans. Some recent additions to the class include propaquizafop, quizalofop-P, metamifop and pyriftalid.
FLUAZIFOP-BUTYL (Fusilade®)
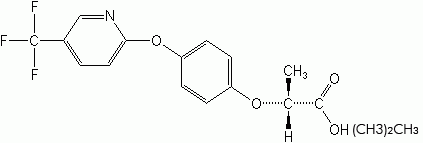
Phenoxys
2,4-D was introduced in 1944 as the first of the “phenoxy herbicides,” “phenoxyacetic acid derivatives,” or “hormone” weed killers. These were highly selective for broadleaf weeds and were translocated throughout the plant. 2,4-D provided most of the impetus in the commercial search for other organic herbicides in the 1940s. Several compounds belong to this group, of which 2,4-D and 2,4,5-T are the most familiar. Other important compounds in this group are 2,4-DB, MCPA, and silvex.
2,4-D
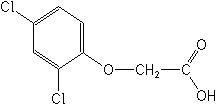
The phenoxy herbicides have complex mechanisms of action resembling those of auxins (growth hormones). They affect cellular division, activate phosphate metabolism, and modify nucleic acid metabolism.
2,4-D, MCPA, and 2,4,5-T have been used for years in very large volume worldwide. The latter product, 2,4,5-T, used mainly to control woody perennials, became the subject of extended investigation, particularly because of its use in Vietnam in combination with 2,4-D as Agent Orange. Certain samples were found to contain small amounts of a highly toxic impurity, 2,3,7,8-tetrachlorodibenzo-p-dioxin, commonly referred to as tetrachlorodioxin, or dioxin. Although alterations in manufacturing procedures reduced the dioxin content to minimal levels, 2,4,5-T registrations were cancelled and the product voluntarily removed by the manufacturers in 1985.
Agent Orange was replaced with Agent White, a mixture of 2,4-D and picloram, which was longer lasting and more effective. That same combination is used today (Grazon®P+D, Tordon®RTU) on pastures and rangeland for control of woody species.
2,4,-D has been and continues to be one of the most useful herbicides developed. More than 33 million pounds manufactured in the United States, is used each year in 35 ester and salt forms. In agriculture it is used on cereal, grain crops& and sugar cane for the control of broadleaf weeds, and on rights-of-way, turf and lawns, and in forest conservation programs. The manufacturing process for 2,4-D used in the United States does not result in any level of tetrachlorodioxin contamination. Other members of the phenoxys in wide use are dichlorprop (2,4-DP), the butyric acid form (MCPB), and mecoprop (MCPP).
Ureas
Thousands of ureas, also called substituted ureas, have been tested as herbicides and many are in use today. Fluometuron is illustrated. Others include linuron, diuron and monuron, fenuron-TCA, siduron and tebuthiuron. Most of the ureas are relatively nonselective and are usually applied to the soil as preemergence herbicides; some have postemergence uses, while others are active when foliar applied. The ureas are strongly absorbed by the soil, then absorbed by roots. Their mechanism of action is to inhibit photosynthesis—the production of plant sugars—and, indirectly, to inhibit the Hill reaction. Despite the number and age of this group new ones continue to make an appearance. Two recent additions include isoproturon and cumyluron, the latter of which though structurally a urea has an unknown mode of action. Neither is used in the U.S. There are yet a larger number of ureas not registered for use in the U.S.
FLUOMETURON (Cotoran®)
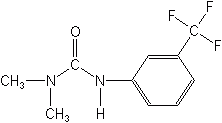
Phosphono Amino Acids
These are foliar applied, translocated herbicides that interfere with normal plant amino acid synthesis. They are more effective against grasses than broadleaf weeds, and are considered to be nonselective foliar compounds with no soil activity. They penetrate rather slowly, thus rainfall shortly after application can reduce effectiveness. Found here are glyphosate, glufosinate, fosamine and glyphosate trimesium.
GLYPHOSATE (Roundup®)
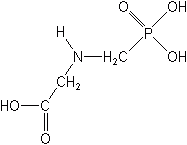
Glyphosate, now the most widely used pesticide in the U.S. and in the world is a nonselective, nonresidual, postemergence material. It is recognized for its effectiveness against perennial, deep-rooted, grass and broadleaf weeds, as well as woody brush problems in crop and noncrop areas. It is a translocated, foliar-applied herbicide that can be applied at any stage of plant growth or at any time of year, with most types of application equipment, including the new wick, roller, and wiper devices. Its mechanism of action appears to be the inhibition of the synthesis of aromatic amino acids, which results in the inhibition of nucleic acid metabolism and protein synthesis. There are a number of salt forms registered for glyphosate.
The growth of glyphosate was dramatically enhanced starting in the mid-1990s by the creation and marketing of several crops (corn, cotton, soybeans, canola with others planned) bioengineered to render them tolerant to applications of glyphosate. This technological advance allows farmers to spray the non-selective herbicide onto these crops to achieve excellent weed control without significant crop damage. Although similar traits were introduced into certain crops for other herbicides including imadazolinones and glufosinate these have not matched the commercial success enjoyed by glyphosate.
Glufosinate is used primarily for grass weeds in orchards and vineyards as a postemergence herbicide. Surprisingly, it is used as a desiccant on potatoes, and for weed control in minimum tillage systems. Fosamine controls bud growth in woody species in noncrop areas, and glyphosate trimesium (Touchdown®) is the newest of the lot, appearing in 1988.
Phthalic Acids
Only two compounds of significance fall into this minor class. They are chlorthal (DCPA; often sold as the dimethyl ether which is called chlorthal-dimethyl), developed in 1960, and endothall, which appeared in 1965. Chlorothal is a quality crabgrass herbicide, but is also highly effective against most grass and broadleaf weeds in vegetables. Endothall is used in sugar beets for a narrow range of weeds, as an aquatic herbicide, as an algicide, and as a desiccant for cotton, potatoes, alfalfa and clover. Its low toxicity to fish makes it an ideal aquatic herbicide, and an outstanding example of environmental protection through pesticide selectivity.
DCPA, CHLORTHAL (Dacthal®)
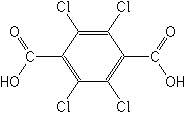
Pyridazinones
Because these herbicides inhibit the formation of chlorophyll, the green pigment in plants, the weeds soon lose their color following uptake, and thus appear bleached. They are also referred to as being carotinoid biosynthesis inhibitors. These newer herbicides control grasses and broadleaf weeds, are compatible with the bipyridiliums and a wide range of preplant soil incorporated preemergence herbicides.
Pyrazon (formerly chloridizon), used almost exclusively on sugar beets, is a selective, pre- and postemergence herbicide for broadleaf weeds and is unusual for the group in being a photosystem II inhibitor. Norflurazon is used as a preemergence herbicide for control of annual grasses and certain broadleaf weeds in cotton, soybeans, and vine and deciduous fruit crops. Fluridone is an aquatic herbicide used in ponds, drainage and irrigation canals, lakes and reservoirs, for the control of most aquatic weeds at any stage of growth.
NORFLURAZON (Solicam®, Evital®)
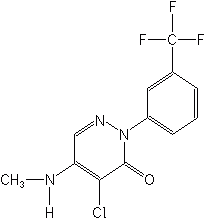
Fluorochloridone (Racer®), an experimental pre-emergence herbicide for grass and broadleaf weeds in a wide range of crops. Clomazone (formerly dimethazone) is a pre-emergence or preplant incorporated herbicide for grass and broadleaf weeds in soybeans, cotton and vegetables.
Oxadiazon is used for weed control in dry-seeded rice, and for turf and ornamentals. Among the newest family members are beflubutamid, picolinafen and oxadiargyl. The first is an experiment being evaluated for use on small grains. Oxadiargyl is not used in the U.S. Oxadiargyl like oxadiazon (Ronstar®) is an inhibitor of protoporphyrinogen oxidase. Picolinafen (Pico®) has potential uses on small grains and is an inhibitor of phytoene desaturase.
Carboxylic Acids (formerly Pyridinoxy and Picolinic Acids)
The carboxylic acid herbicides like their benzoic acid relatives are identified by the carboxylic acid radical attached to the benzene ring. More specifically, this COOH group is usually referred to as the carboxyl group. In this group are picloram, triclopyr, fluoroxypyr, clopyralid and quinclorac. These are hormone herbicides that translocate in both the phloem and xylem. They are ideal for control of perennial broadleaf weeds and brush, and have both soil and foliar activity. Picloram is long-lived in soil, while triclopyr and clopyralid are much less persistent. Their mode of action is that of the synthetic auxins, acting like the natural plant hormone indoleacetic acid, and like their kindred phenoxy-, and benzoic-acid herbicides.
PICLORAM (Tordon®)
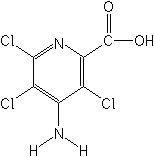
Picloram, combined with 2,4-D to form Agent White, was used in Viet Nam as a replacement for Agent Orange (2,4-D and 2,4,5-T). That same combination is used today on pastures and rangeland for the control of woody species under the proprietary names of Grazon®P+D, Pathway®, and Tordon®RTU.
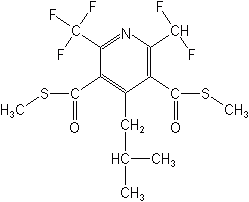
Pyridines
Pyridine is a benzene ring with one of the carbons replaced by a nitrogen, as in the illustration. There are several substituted pyridines used as herbicides. In this group are dithiopyr (Dimension®) and thiazopyr (Visor®). Dithiopyr is a selective pre- and postemergence material, used only on turf to control a wide variety of grass weeds. It is frequently formulated with other herbicides and on fertilizers. Thiazopyr is a selective preemergence compound that works well for practically all grass weeds, on a wide variety of crops, including citrus, cotton, corn, peanuts, soybeans and potatoes. They appear to be microtubule assembly inhibitors.
DITHIOPYR (Dimension®)
Sulfonylureas
The sulfonylureas are meristematic inhibitors with both foliar and soil activity which control broadleaf weeds better than grasses. They have very specific activity with application rates often being less than one-fourth ounce (7 grams) per acre. They range in persistence from very long to moderate, and have very low mammalian toxicity. The mode of action for the sulfonylureas is the inhibition of branched chain amino acid biosynthesis through the inhibition of an enzyme called acetolactate synthetase (ALS).
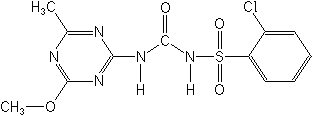
In this group belong chlorsulfuron, chlorimuron-ethyl, metsulfuron-methyl, and sulfometuron-methyl. Chlorsulfuron appeared in 1981, and works well for broadleaf weeds in wheat, barley, oats and non-crop weed control. Wild oats are tolerant to this material. Chlorimuron-ethyl controls broadleaf weeds in soybeans as a postemergence herbicide. Metsulfuron-methyl is used in the same role as chlorsulfuron. Sulfometuron-methyl is used only on noncrop areas for nonselective weed control as a broad spectrum pre- and postemergence herbicide. It can also be used in pine forests to control woody tree species. Thifensulfuron-methyl is a selective postemergence material for broadleaf weeds and wild garlic in small grains. Halosulfuron-methyl (Battalion®, Permit®) is a selective pre- and postemergence compound for corn, sorghum, rice, sugarcane and turf.
CHLORSULFURON (Glean®)
Additional older sulfonylureas are bensulfuron (Londax®), nicosulfuron (Accent®), triasulfuron (Amber®, Logran®) and primisulfuron-methyl (Beacon®, Tell®).
The newer sulfonylureas are amidosulfuron (Eagle®, Pursuit®), azimsulfuron (Gulliver®), cyclosulfamuron (Invest®), ethametsulfuron-methyl (Muster®), ethoxysulfuron (Sunrise®), flupyrsulfuron-methyl (Lexus®), foramsulfuron (Equip®, Option®), iodosulfuron-methyl (Husar®), mesosulfuron-methyl (Mesomaxx®), oxasulfuron (Dynam®, Expert®), prosulfuron (Peak®), rimsulfuron (Matrix®, Shadeout®), sulfosulfuron (Maverick®, Sundance®), tribenuron-methyl (Express®), trifloxysufuron (Brawn®, Enfield®), triflusulfuron-methyl (Debut®, Upbeet®), and tritosulfuron (Corto®). Foramsulfuron and Iodosulfuron-methyl were registered by the U.S. EPA in 2002 while trifloxysulfuron was registered in 2003. For the past two decades this has been among the most fertile groups for new herbicide discovery of all herbicide classes, and though now somewhat mature may continue to receive new candidates from the major U.S. and Japanese manufacturers.
Thiocarbamates
The thiocarbamates contains sulfur (from the Greek word theion) and are derived from the hypothetical thiocarbamic acid.
The thiocarbamates are selective herbicides marketed for weed control in croplands. The thio- and dithiocarbamates have exceptional vapor pressures, are quite volatile, and must be incorporated in the soil at time of application. They inhibit the development of seedling shoots and roots as they emerge from the weed seeds. Consequently, they are all used as either preplant or preemergence, soil-incorporated herbicides.
Examples of thiocarbamates are EPTC and pebulate. EPTC is used mostly to control annual and perennial grassy weeds, including nutgrass, johnsongrass, and quackgrass. Pebulate is effective against both grassy and broadleaf weeds in tobacco, and tomatoes. Also included among the thiocarbamates are thiobencarb, butylate, cycloate, molinate, and vernolate. Thiobencarb is the most widely used rice herbicide Worldwide. It is a preemergent and early postemergent compound, for grasses and broadleaf weeds in rice, exclusively.
EPTC (Eptam®)
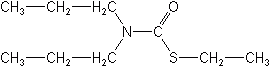
Molinate is effective for barnyard grass control in rice. Cycloate is used mostly in sugar beets, table beets, and spinach to control annual and perennial grasses and broadleaf weeds. Vernolate is effective for control of most grass weeds and some broadleafs in soybeans and peanuts. Butylate is incorporated preplant to control grass weeds in corn.
Two older thiocarbamates not illustrated, are diallate (Avadex®) and triallate (Far-Go®). Diallate (discontinued) is for wild oats and other grass weeds in a wide variety of crops. Triallate controls only wild oats in small grains.
Triazines
The triazines, which are six-member rings containing three nitrogens (the prefix tri- means “three”) and azine (a nitrogen-containing ring) make up the heterocyclic nitrogens.
Triazines are strong inhibitors of photosynthetic electron transport. Their selectivity depends on the ability of tolerant plants to degrade or metabolize the parent compound (the susceptible plants do not). Triazines are applied to the soil primarily for their postemergence activity. There are many triazines on the market today, a few of which are illustrated.
Atrazine was estimated by EPA to have been the most heavily used pesticide in the U.S. in 1999, used at the annual rate of about 80 million pounds, primarily on corn. (Donaldson et. Al., 2002). In 2003 EPA concluded a key milestone in a special review of atrazine use that had gone on for many years. Though many uses of the product will continue a number of restrictions in use rates and pattern will apply in order to reduce the probability of atrazine residues moving to ground or surface water.
ATRAZINE (Aatrex®)
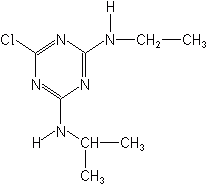
In a 1996 agreement with EPA uses for cyanazine were phased out by the manufacturer. Other important triazines not illustrated, are prometryn (Caparol®), ametryn (Evik®), and terbutryne (Igran®).
The triazines are used selectively in certain crops and nonselectively in others. They are used in greatest quantity in corn production and nonselectively on industrial sites.
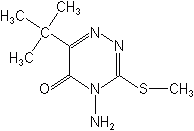
Triazinones
The triazinones consist of two herbicides, metribuzin and hexazinone. Metribuzin is used on soybeans, wheat, sugarcane, and a few vegetables. Hexazinone is used in the control of annual, biennial, perennial weeds and woody plants on noncropland. It is sometimes applied as pellets or gridballs to control woody plants that fall to the ground through the dense foliage of conifer plantings e.g., Christmas trees.
METRIBUZIN (Sencor®)
Triazoles
The oldest and only member of this group is amitrol, which became embroiled in the historic "cranberry incident" of 1959. This culminated in the addition of the Food Additive Amendment of 1958 (also called the Delaney clause) to the Federal Food, drug, and Cosmetic Act. In essence, this amendment stated that no residue of any carcinogen (cancer-producing agent) would be permitted in a food crop.
Uracils
Below is illustrated the uracil nucleus, and on it are substituted several chemical groups or moities, to form the herbicides bromacil, terbacil, and a third sold only in Europe, lenacil. Bromacil is a pre- and postemergence herbicide, for noncrop usage, pineapples, and citrus. It controls a very wide spectrum of grass and broadleaf weeds when applied early in the growing season, and must be carried into the root zone by moisture to be absorbed by the roots. Terbacil controls most annual grasses and broadleaf weeds when applied as a selective, preemergence herbicide. As with bromacil, moisture is required to move the material into the weed germination zone by sprinkler irrigation or rainfall. Terbacil has an extended soil residual life and non-tolerant crops should not be planted in treated fields for two years, nor should terbacil be used in sandy soils or those with less than 1% organic matter.
BROMACIL (Hyvar®)
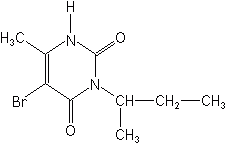
The uracils inhibit photosynthesis by blocking the Hill reaction, as do the ureas and triazines. They are usually applied to the soil and move in the transpiration stream.
New Novel Herbicide Classes
A number of new products representing structures that do not easily fit into those classes covered above have appeared and are discussed below by class.
Benzoylcyclohexanediones- The three members of this class are mesotrione (Callisto®), sulcotrione (Mikado®) and benzofenap (Yukawide®). Mesotrione is the only of this group used in the U.S. and it was first registered in 2001 for use in corn to control broadleaf weeds and crabgrass. Sulcotrione is used in maize and sugarcane and benzofenap in rice. All of these materials are inhibitors of 4-hydroxyphenyl-pyruvatedioxygenase (4-HPPD).
MESOTRIONE (Callisto®)
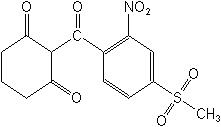
N-phenyl-phthalimides-Cinidon-ethyl (Lotus®, Bingo®) is used to control broadleaf weeds in small grains outside the U.S. This product acts by inhibiting protoporphyrinogen (PPO).
CINIDON-ETHYL (Lotus®)
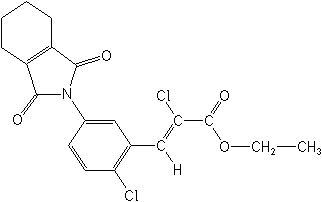
Oxazolidinediones- Pentoxazone (Wechser®) is the only member of this class, at present. It is used on rice and at present only in S. Korea. It is an inhibitor of PPO.
PENTOXAZONE (Wechser®)
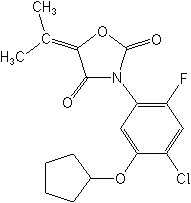
Phenyl-pyrazoles-The only member of this class is pyraflufen-ethyl (Ecopart®) and is now used outside the U.S. but has pending uses for broadleaf control in cotton, sugarcane, wheat, corn, potato and soybean. It is a PPO inhibitor.
Pyrimidindiones-Similar to the triazolinones class butafenacil (Rebin®, Inspire®) is the only member. It is used for weed control in grapes, nut crops, pome and stone fruits and also as a cotton defoliant. Its action is as a PPO inhibitor. Butafenacil was registered by EPA in 2003.
BUTAFENACIL (Inspire®)
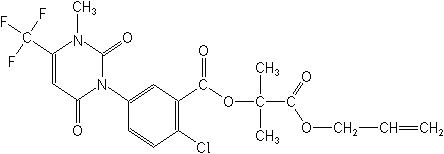
Sulfonyamino-carbonyl-triazolinones- The two new entrants in this class are flucarbazone (Everest®) and propoxycarbazone (Attribute®, Olympus®) each of which exists as sodium salts. Each of these products is used in small grains to control grassy annual and some broadleaf weeds. Flucarbazone is registered on wheat in Canada and the U.S. Both products are inhibitors of acetolactate synthetase like the sulfonylureas.
FLUCARBAZONE (Everest®)
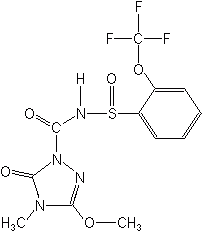
Tetrazolinones-fentrazamide (Lecs®) is structurally reminiscent of the amide and chloroacetamid herbicides and is the only member of this class. It is a low toxicity material which is used to control barnyardgrass and annual sedges in rice in Japan and Korea. Like many of the amides and chloroacetamides it acts by inhibiting very long chain fatty acid synthesis,
FENTRAZAMIDE (Lecs®)
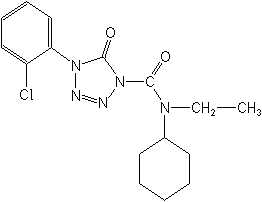
Triazolinones- Amicarbazone (Dinamic®) is used to control broadleaf weeds in corn and soybeans and perhaps sugarcane. It is an inhibitor of photosystem II and is slated for a maize registration in the U.S. as early as late 2004.
AMICARBAZONE (Dinamic®)
Unclassified or Miscellaneous
Three herbicides have recently appeared that do not fit any particular established structural class. There are flufenpyr-ethyl (Axiom®), oxoziclomefone (Homerun®, Patful®) and benzobicyclon (Showace®). Flufenpyr-ethyl was registered by EPA in 2003 for corn, soybeans and sugarcane. It is a PPO inhibitor. Oxoziclomefone and benzobicyclon are both for use on rice in Japan. Their modes of action are not yet known.
Soaps
The principal value of soaps is in their capacity to reduce the surface tension of water, thus causing the guard cells surrounding the stomata to collapse. These in turn close or clog the stomata, the oxygen:carbon dioxide-exchange organs of the plants, causing death to the surrounding cells.
Soaps are the alkali salts of fatty acids. Both soft soaps (potassium salts) and the hard soaps (sodium salts) are water soluble, but only the soft soaps are herbicidal. Overall, the most efficacious fatty acid salts are those near the carbon chain length of lauric acid (C12), which include caproic (C10), myristic (C14), palmitic (C16) and stearic acids (C18).
In the late-1980s the first soft soap herbicides were registered. The two major manufacturers are the Ringer Corporation, which produces Safer® Moss & Algae Killer, and the Mycogen Corporation, which produces DeMoss® Moss/Algicide, and Sharpshooter® Herbicide, all aimed primarily toward the home yard and turf market.
Petroleum Oils
The earliest organic herbicides were the petroleum oils, which are a complex mixture of long-chain hydrocarbons. They include alkanes, alkenes, and often alicyclics and aromatics, produced by distillation and refinement of crude oils. The petroleum oils exert their lethal effect by penetrating and disrupting plasma membranes, which is enhanced by temperature and direct sunlight. Because of EPA’s and many local and state governments’ regulations to halt the release of hydrocarbons into the atmosphere, petroleum oils are no longer prominently used as herbicides though they are used as adjuvants in herbicide formulations.
Herbicide Safeners
It has long been known that mixing certain substances with herbicides before application will impart a protective or safening effect on selected crops without substantively affecting the weed control achieved. Few such agents were widely used before the last decade, however. Recently, the search for such substances has increased as producers have sought incremental performance and market advantages for their products.
Herbicide safeners are a diverse collection of compounds and many appear to impart their effects by enhancing the metabolism of the herbicide by the monocotyledonous crops on which the herbicide is used.
A few prominent herbicide safeners include: Cloquintocet-mexyl used with clodinafop-propargyl in Discover® Herbicide to safen the active ingredient on wheat. This safener and the herbicide with which it is used were first registered in the U.S. in 2000. Benoxacor, registered in 1997, increased the tolerance of s-metolachlor (and metolachlor) to corn. Dichlormid increases corn tolerance to thiocarbamate herbicides. Fenchlorazole-ethyl synergizes the activity of fenoxyprop-ethyl in susceptible species including wheat where the main action is to reduce phytotoxicity and growth retardation. Similarly, mefenpyr-diethyl safens fenoxyprop-P-ethyl in small grains. Fluxofenim and oxabetrinil both safen sorghum from metolachlor injury when applied as a seed treatment, particularly when metolachlor is used in combination with a triazine herbicide. Furilazole is an herbicide safener designed for wide range of herbicides on cereals, corn and rice. Finally, isoxadifen-ethyl is a safener for corn which has only very recently appeared.
Biopesticides
In addition to herbicidal agents of synthetic chemical origin, there are also a few so called “biopesticides”, which are natural or semi-natural agents or organisms that can exact a degree of control over specific pests. A number of microorganisms (bacteria, viruses and fungi) and a few insects (e.g., parasitic wasps and the painted lady butterfly) have been evaluated as bioherbicides. In general, the only category that has yielded commercial examples, and these are few, are fungi (mycoherbicides).
The first to be registered was Phytophthora palmivora (Devine®) which induced selective disease on milkweed vine. Another commercial example is Collego®, living spores of the fungus Collectotrichum gloeosporioides F. sp. aeschynomene. It is applied to control coffee weed in rice or northern joint-vetch in soybeans. This fungus requires several weeks to achieve control, and to work at all the crop must be misted or the weather very humid.
Alternaria cassiae, though dropped as a commercial prospect, is also a myco-herbicide. It showed promise in controlling sicklepod in several crops.
Two other developmental myco-herbicides are: Puccinia canaliculata and Xanthomonas campestis for control of yellow nutsedge and annual bluegrass, respectively. Canada is exploring a new mycoherbicide, Phoma macrostoma, for use in turf.
Trangenic Pesticides (Herbicide-Tolerant Crops)
Transgenic organisms are genetically altered by artificial introduction of DNA from another organism. The artificial gene sequence is referred to as a transgene. Plants with such transgenes are also referred to as being genetically modified (GM). Plants that emulate insecticides are those altered to induce insect-resistance (also called plant pesticides or plant incorporated protectants). The other category of transgenic pesticides that we will touch on below is herbicide-tolerant crops.
Herbicide-tolerant crops are genetically engineered to sustain the action of selected herbicides applied directly over the young plants during standard weed control treatments. The significant commercial introduction of herbicide-tolerant crops began in the mid 1990’s and prominent ones include corn, cotton and soybeans (among many others) designed to withstand the action of non-selective herbicides like glyphosate or glufosinate. Such tolerant crops allow broad spectrum weed control and high crop safety. Notwithstanding, there have been a number of crops engineered to induce tolerance to several other herbicides. Examples include bromoxynil, several imadazolinone and sulfonylurea herbicides, among others.
A good review of the history, scientific process and status of such products can be viewed at the following website: (http://www.colostate.edu/programs/lifesciences/TransgenicCrops/).
n.b.: All cited websites were successfully reached on 14 June, 2006. They are subject to change, however, which may require that you search keywords for changed web addresses
Selected References
- Carpenter MJ , Ware GW (2004) Defending Pesticides in Litigation, 14th Ed., West Group, St. Paul, MN. 1054 pp.
- Crop Protection Handbook (2004) Meister Publishing Co., Willoughby, Ohio. Vol. 90, 897 pp.
- Donaldson D, Kiely T, Grube A (2002) Pesticides Industry Sales and Usage, 1998 and 1999 Market Estimates. US EPA, Washington, DC. EPA-733-R-02-001, 34 pp.
- Herbicide Classification of the Weed Science Society of America (1997) Weed Technology 11:384-393.
- Herbicide Handbook (2002) Weed Science Society of America, 8th Ed. (Vencill WK, Ed), Lawrence, Kansas. 493 pp.
- Pesticide Manual (2000) A World Compendium, 12th Ed., Tomlin CDS (Ed.). British Crop Protection Council. Farnham, Surrey, UK. 1250 pp.
- Ware GW, Whitacre DM (2004) The Pesticide Book, 6th Ed. Meister Media Worldwide. Willoughby, Ohio. 488 pp.