Kathy L. Flanders
Department of Entomology
Auburn University
Auburn, AL 36849-5629
Edward B. Radcliffe
Department of Entomology
University of Minnesota
St. Paul, MN 55108-6125
Alfalfa, Medicago sativa, is the most important forage crop world-wide. In the US it is grown on approximately 10 million hectares (25 million acres) and has an on-farm value of over $5 billion. Minnesota, and the adjacent states of South Dakota, Iowa, and Wisconsin produce 1/4 of US alfalfa. Alfalfa is unique among field crops in that it represents a relatively long lasting (typically 3-4 years), perennial ecosystem that exists in virtually all parts of the country. Alfalfa is adapted to a wide range of climatic conditions and thrives in cool, humid environments and in most irrigated arid areas. In the Midwest, alfalfa is commonly grown in alfalfa-corn-soybean systems, in the West, often in alfalfa-sugarbeet systems, and in the East often in alfalfa-orchard or alfalfa-pasture systems.
Alfalfa supports a diverse arthropod fauna. At least 1,000 species have reported from alfalfa in the US, with perhaps 100-150 of these causing some degree of injury. Few of these, however, can be described as key pest species, the rest are of only local or sporadic importance, or are incidental herbivores, entomophagous (parasites and predators), or pollinators.
Annual dollar loses due to insect pests in alfalfa runs into the hundreds of millions. Yield reductions in the range of 10-15% are typical estimates (USDA 1965), but are probably low because the question of forage quality has usually not been considered. Alfalfa does not in the usual sense have a market value because production is usually for use as livestock feed by producer. True value is determined by feed value, e.g., milk production.
With homopterous pests it is often not possible to demonstrate dry weight yield loss (the bulk of the weight being stems), but one can still have significant leaf drop and loss of protein and in vitro dry matter digestibility (IVDMD). Insects may also impair the persistence (e.g., winter kill) and longevity of alfalfa stands.
The key pests include alfalfa weevil, Hypera postica (a species that we consider includes Egyptian alfalfa weevil, H. brunneipennis), as well as a complex of homopterous pests, the most important of which are potato leafhopper, Empoasca fabae, spotted alfalfa aphid, Therioaphis maculata, pea aphid, Acyrthosiphon pisum, blue alfalfa aphid, A. kondoi, threecornered alfalfa hopper Spissistilus festinus, and meadow spittlebug, Philaneus spumarius. In seed production, serious losses can be caused by mirids, especially Lygus hesperus, and L. elisus, and the clover seed chalcid, Bruchophagus roddi (Eurytomidae). In the South, armyworms, Spodoptera spp., (Leptidoptera: Noctuidae) and webworms, Loxostege spp. and Achyra spp. (Lepidoptera: Pyralidae) are important pests. The alfalfa snout beetle, Otiorhynchus ligustici (Coleoptera: Curculionidae), a serious pest, is thus far limited in distribution to northern New York State and southern Ontario. Blister beetles, Epicauta spp. (Coleoptera: Meloidae), do not injure alfalfa, but are of concern because they are poisonous to livestock.
Insecticide use on alfalfa in northern states has generally not been great. Typically, 1-5% of the Minnesota crop is sprayed in any given year. In some parts of the country, because of greater pest pressure, insecticidal treatment is more general and sometimes routine. In Georgia, 85% of alfalfa is treated with insecticides each year. Overall, the crop ranks 18th out of 87 crops in pounds of pesticide (insecticides, herbicides, and fungicides) applied in the U.S. (10.8 million pounds out of 886 million pounds used annually). Much of insecticide applied to forage alfalfa is directed against alfalfa weevil. In recent years, biological control agents and host plant resistance have diminished the impact of this insect and accordingly the need for chemical control.
Alfalfa has several attributes which make it an excellent candidate for the application of integrated pest management approaches. Cows tend to be tolerant of insect damage and/or insect parts in their diet so there is no reason not to establish realistic economic thresholds. Moreover, alfalfa is for the most part, quite tolerant of moderate pest densities. This permits greater reliance on alternative control methods. Because the crop is semi-permanent, and supports a diverse arthropod fauna, biological control has considerable potential. Biological control agents such as the parasitoids Aphidius ervi and Praon pequodorum, and generalist predators such as coccinellid beetles, keep pea aphid populations below economic injury levels in much of the United States. Introduced parasitoids are important biological control agents of alfalfa weevil.
Alfalfa offers considerable opportunities for cultural manipulations. The crop is different from many in that it is typically harvested 2-4 times in the northern US, and 6-10 times in the southern US. Harvesting can reduce pest populations by 95-98%. In Minnesota, delay of first harvest until 1/10th bloom (approximately June 15) allows sufficient time for potato leafhopper oviposition to occur. These eggs (inserted into the stems) are then harvested with the crop, virtually eliminating that generation. Conversely, harvesting alfalfa early, when alfalfa weevil populations are near peak, can eliminate the need for an insecticide application. Burning and mowing have been used to kill overwintering alfalfa weevil eggs. Planting alfalfa in a grass mixture can reduce pressure by both potato leafhopper and alfalfa weevil. Grazing tolerant alfalfa varieties have been developed that enable cattle to continuously graze alfalfa. In future, grazing may become a viable cultural management practice.
Host plant resistance to spotted alfalfa aphid, is common in alfalfa cultivars, thanks to efforts by plant breeders. Development of alfalfa cultivars with resistance to alfalfa weevil proved one of the most difficult breeding problems ever undertaken in alfalfa. A program of recurrent selection was undertaken by researchers at the USDA Beltsville Laboratory. Over 2 million seedlings were exposed to massive weevil infestations. From this material, selections were made that eventually to the cultivar, Team, released in 1969. Team has partial resistance to weevil. This resistance is based on tolerance and some degree of antibiosis. In areas where Team is well adapted it has been credited with contributing significantly to reductions in weevil pest pressure. Germplasm releases with glandular-haired trichomes derived from crosses with wild Medicago species, e.g., KS 224 and KS108GH5, possess resistance to potato leafhopper and alfalfa weevil. "Hairy" alfalfa varieties derived from these lines should be available soon.
Major Insect Pests of Alfalfa









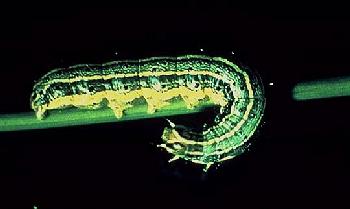



The Alfalfa Weevil and its Natural Enemies
Alfalfa weevil was first discovered in the US in 1904 near Salt Lake City. It is assumed it was probably introduced from southern Europe. For the next 50 years, alfalfa weevil gradually expanded it's range until it occupied most of the region west of the 100th meridian. A second introduction, which for many years was considered to be a closely related but different species, the Egyptian alfalfa weevil, invaded Arizona and southern California in 1939. That insect apparently has remained confined to the hot valley regions of the Southwest. In 1952, alfalfa weevil was discovered near Baltimore and from there spread rapidly throughout the eastern, southern, and midwestern states. The eastern population weevil proved much more aggressive than the western weevil and caused major economic losses. In the southeastern United States alfalfa was virtually eliminated as an agricultural crop. In 1970, alfalfa weevil (eastern population) was discovered in Minnesota and within 3 years had moved across the state. Minnesota populations have, however, remained at subeconomic levels. The eastern and western populations have now merged west of the Mississippi River, and alfalfa weevil occurs in all 48 contiguous states.
Until recently, eastern alfalfa weevil and western alfalfa weevil were considered to represent distinct "strains" (= subspecies) of H. postica, while Egyptian alfalfa weevil was considered to be a different species, H. brunneipennis. The eastern and western populations are not completely isolated, but differ in details of biology, e.g., the eastern weevil returns from summer aestivation sites to the alfalfa in the fall whereas the western weevil does not return until spring. Entomologists now recognize that eastern alfalfa weevil is actually more closely related to the Egyptian alfalfa weevil than it is to the western alfalfa weevil. Comparison of isoenzymes (Hsiao) tends to support this view.
In the old world, alfalfa weevil is a species having a remarkable range of distribution, occurring from Scandinavia to Egypt and Iran, therefore, it is not surprising that distinct biotypes exist. Even within the populations there is considerable biological plasticity, e.g., in the south, alfalfa weevil may reach sexual maturity and begin egg laying in the fall, whereas in the north, egg laying is confined to the spring. This makes alfalfa weevil much more difficult to control in the southern portion of it's range.
Biological control of the weevil was first attempted in 1911 with the introduction of the icheumonid parasite, Bathyplectes curculionis from Italy. The parasite proved moderately effective in cooler climates, but has been less successful in hot climates, e.g., San Joaquin valley. B. curculionis also attacks Egyptian alfalfa weevil, but initial introductions were ineffective, presumably due to the hot weather of habitats favored by Egyptian alfalfa weevil and encapsulation of the parasites eggs by that host. Introduction of a new strain of B. curculionis from Iran was attempted to overcome these limitations, but success was not achieved until the introduction of a second species, B. anurus.
Similarly in the eastern US, alfalfa weevil tends to encapsulate B. curculionis reducing the parasites effectiveness. Attempts have been made to establish additional parasitic species against the eastern alfalfa weevil and at least 5 of these are now widely distributed: B. anurus, Oomyzus incertus, Microctonus aethiopoides and M. colesi and Patasson luna. Recently, the egg predator Peridesmia discus has been recovered, years after the initial releases were made.
In areas where 3 or more parasites are established, alfalfa weevil is seldom of economic importance, even in areas once requiring multiple applications of insecticide. Often as B. anurus has become established, it has tended to displace B. curculionis and O. incertus. This process is most likely in situations where the fungal pathogen Zoophthora (=Erynia, =Entomophthora) phytonomi causes epizootics killing large numbers of alfalfa weevils. Bathyplectes anurus has an advantage when fungal epizooticss occur because it attacks alfalfa weevil larvae slightly earlier in the season than does B. curculionis and appreciably earlier than O. incertus. Thus, B. anurus is more likely to have completed its development before host larvae begin to die. Also, B. anurus has only one generation per year whereas B. curculionis and O. incertus tend to be multvoltine. Bathyplectes curculionis has been in Minnesota since arrival of the alfalfa weevil in 1970, but B. anurus has only be established since 1991. To date, there is no evidence that B. anurus has displaced B. curculionis in Minnesota, but B. anurus now predominates in Iowa.
Zoophthora phytonomi was first identified from alfalfa weevil larvae in Ontario in 1973. The pathogen, is similar, but is presumed different from that occurring in the clover leaf weevil, Hypera punctata. The pathogen is now the key natural mortality factor in much of eastern Canada and the eastern and southern US.
Important Biological Control Agents




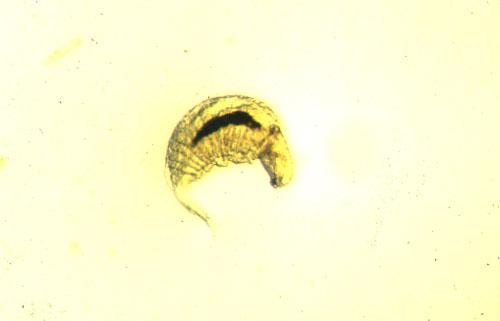




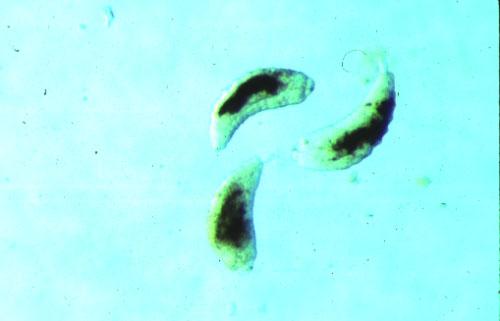


Potato Leafhopper and Pea Aphid
Much of our own efforts in Minnesota have concerned the establishment of economic thresholds for two insects, potato leafhopper and pea aphid. We effected gradients in densities of either insect alone or in combination by application of selective insecticidal treatments (i.e., by varying the rates, timing, or choice of insecticide) and by the management techniques used on the alfalfa bordering treatment plots (i.e., strip-cutting, earlier or later harvest dates, or by treating with insecticide). For example, in our experiments, alfalfa plots bordered by uncut alfalfa had 4 times as many potato leafhopper adults and 10 times as many nymphs as alfalfa having borders cut on the same schedule as the plot itself.
In this way, we were able to establish and maintain gradients of pest pressure in the plots. We measured a number of yield parameters including dry matter yield, stem height, maturity, % protein, and in-vitro dry matter digestibility. Each was found to be influenced by pest densities. From this we were able to compute economic injury levels and economic thresholds. To take into account the effects on forage quality we computed these values on the basis of an equivalent (substitute) ration of corn and soybean meal at prevailing prices.
For potato leafhopper, economic injury levels were found to be in the range of 30 leafhoppers/0.48m2/sample week for vacuum-sampling or 37 leafhoppers/25 180-degree sweeps/sample week. The critical time at which potato leafhopper is most capable of causing plant injury was found to be on the regrowth (stubble) immediately following harvest. Economic thresholds (=action thresholds) ranged from 0.3 to 0.5 leafhoppers/pendulum sweep for alfalfa at 5-17 cm regrowth.
For pea aphid, Cuperus et al. calculated economic injury levels were found to be in the range of 3,850 aphid-days/vacuum sample, 3,950 aphid-days/sweep, or 114 aphid-days/stem, respectively. The critical time for control appeared to be 2 weeks prior to harvest. Economic thresholds 2 weeks before harvest were 58 aphids/vacuum sample, 70 aphids/sweep, or 1.2 aphids/stem, respectively.
From these data it seems evident that potato leafhopper and pea aphid are very different in their potential to cause injury. Very low numbers of potato leafhopper may cause economic loss, whereas the threshold for pea aphid is very high.
In a subsequent study, Stucker and Radcliffe, calculated an economic injury levels of 284 cumulative pea aphid-days assuming a control cost of $16/ha and $110/ton hay. This threshold is 80% lower than reported by Cuperus et al., but was determined in experiments with much lower pea aphid pressure. We do not have enough data on pea aphid to know the relationship between economic injury levels and aphid densities, but it may be that the relationship is curvilinear, as is the case with potato leafhopper, in which case there would be a flattening of the response curve at high pest densities.
Regional Research Projects on Alfalfa Insect Pest Management - Past and Present
The Huffaker alfalfa subproject concentrated primarily on the alfalfa weevil in the East and Midwest and Egyptian alfalfa weevil in the west. Development of a descriptive model of the alfalfa plant was undertaken at Cornell (Fick), development of the model for alfalfa weevil and it's principal parasite, B. curculionis was done at Illinois (Ruesink). Optimization models for alfalfa pest management were developed at Cornell (Shoemaker). The research on Egyptian alfalfa weevil was done at the University of California (Regev and Gutierrez).
The exercise was said to have had 3 principal benefits: 1) it helped researchers understand how the crop/pest system operated, 2) it identified research needs (deficiencies), 3) it provided a useful mimic of the system permitting derivation of "dynamic" economic thresholds. The thresholds are described as dynamic because they vary with the stage of regrowth and the time of year.
From the time of its discovery in the east, alfalfa weevil so dominated the attentions of forage crop entomologists that other pests received only scant attention. With the eventual successes achieved in reducing the pest status of weevil the need to consider other pests once again became apparent. This finally lead, in 1974 to the formation of a regional alfalfa pest management project, NC-149 (administered as a North Central Regional Project, but actually national in scope) to consider the alfalfa pest complex). From 1979-1988, this group concentrated its attentions on the homopterous pests of alfalfa, i.e., potato leafhopper, meadow spittlebug and the 3 aphid species, pea aphid, spotted alfalfa aphid, and blue alfalfa aphid. In 1989, the focus of this project was switched to spatial dynamics of leafhoppers, and renumbered as NC-193. This was due to an increased recognition of yield losses due to potato leafhopper on alfalfa.
Insect Pest Management Programs
A number of alfalfa pest management programs have been implemented. The first was the Indiana Action Pest Management Program. This involved regular scouting of participating growers fields, entry of the survey data into a computer terminal for processing on a central main-frame computer. The resulting output (recommendations) offered the grower a choice of management alternatives depending on the stage of plant growth, intended cutting date, and possible insecticidal applications. Today, most states have some form of insect pest management program for alfalfa. These range from recommendations on how to scout for alfalfa weevil to expert systems that model the alfalfa plant and its associated pest.
Key pests in the alfalfa ecosystem differ in different regions of the country. Management of the crop itself varies tremendously: number of harvests per year ranges from 2-10, and the crop may or may not be irrigated. Alfalfa grown for seed has its own particular pest problems. Because of this, integrated pest management strategies differ. Discussion of management strategies for Minnesota, northern New York State, Pennsylvania, Alabama, Oklahoma, and Arizona follow. Special thanks to Michael Rethwisch, Richard Berberet, and Elson Shields, and Arthur Hower for information on alfalfa IPM in their regions.
Minnesota
Key pest of alfalfa in Minnesota is the potato leafhopper. Pea aphid and alfalfa weevil populations seldom reach economic densities, due to a combination of biological control agents and adverse climatic factors. Potato leafhopper is usually a pest of second and third crop alfalfa.
Northern New York
In northern New York state and southeastern Ontario, Canada, alfalfa snout beetle significantly reduces the productivity of alfalfa and clover stands. Alfalfa snout beetle larvae prune lateral roots of alfalfa and commonly sever the tap root. Alfalfa snout beetle-damaged plants often senesce due to dehydration and die during the course of winter causing dramatic stand losses which are often mistaken for winter-kill. Insecticide applications applied in the spring against invading alfalfa snout beetle adults don't reliably control alfalfa snout beetle infestations. Within the alfalfa snout beetle infested area (about 200,000 hectares), dairy farmers are often forced to rotate alfalfa or clover with crops that are not hosts for alfalfa snout beetle (e.g., corn, soybeans, wheat) or substitute legume forages with less cost effective grass hay crops. Crop rotation may reduce overall populations of alfalfa snout beetle, but fields must be fall plowed on a four-year (third production year) schedule and on an areawide basis to prevent rapid reinvasion of alfalfa snout beetle from neighboring fields. For most farmers, this practice is too costly to adopt. Insect-attacking nematodes show promise for management of this pest in future.
Pennsylvania
Alfalfa is harvested 3-4 times per year in Pennsylvania. Key pests are potato leafhoper and alfalfa weevil. Clover root curculio, Sitona hispidulus, significantly contributes to alfalfa stand decline.
PLEX is an expert system designed to help manage potato leafhoppers in Pennsylvania. PLEX models the development of the alfalfa crop and the potato leafhopper, and makes recommendations ranging from cutting management to insecticide applications.
Root feeding by clover root curculio larvae penetrates the phloem and xylem in the lateral and tap roots. These feeding scars directly interfere with the alfalfa root's potential for water and nutrient transport and allow the invasion of pathogens responsible for root rot and wilt. Growers throughout the Northeast lose an estimated $400 million per year from the clover root curculio/fusarium complex due to reduced crop productivity and subsequent increases in replanting costs. Insecticides are not recommended for clover root curculio management since insecticide applications targeted against adult clover root curculio could interfere with established parasites and natural enemies of the alfalfa weevil. Furthermore, insecticide applications are ineffective against the subterranean or larval stage of clover root curculio.
Alabama
Alfalfa is typically harvested 5 times per year in northern Alabama and 10 times in southern Alabama. Before the introduction of alfalfa weevil in 1958, 50,000 A of alfalfa was grown in Alabama. The weevil, and the availability of cheap nitrogen sources, caused alfalfa acreage to plummet in the state in the early 1960's. Interest in growing alfalfa has slowly increased, until approximately 16,000 A of alfalfa are grown in Alabama today. Key pests are alfalfa weevil, three cornered alfalfa hopper, Spodoptera sp. armyworms, and webworms. Potato leafhopper may also cause economic losses, particularly in northern Alabama. Biological control agents of alfalfa weevil are unable to keep the population in check in most years. It is recommended that alfalfa fields be scouted weekly for alfalfa weevil and pea aphid in from March - April, and then bi-weekly thereafter for three-cornered alfalfa hopper and Spodoptera armyworms. Fall armyworm, beet armyworm, corn earworm, yellow-striped armyworm, and green cloverworm are generalist feeders that can colonize alfalfa. They typically occur together. Thresholds are based on number of 1 inch caterpillars of any kind.
Southwestern Arizona
Alfalfa is harvested 10-12 times per year in southwestern Arizona. It is grown exclusively under irrigation. In Arizona, insects are managed on a cutting-by-cutting basis. Higher quality spring alfalfa is more valuable than mid-summer alfalfa. Economic thresholds and injury levels are lower for the spring crops than for the summer crops. The Egyptian Alfalfa weevil is the most important pest of alfalfa in Arizona. Empoasca leafhoppers and beet armyworms occur later in the season. In many cases, it may not be economical to treat, because these cuttings of alfalfa are of lesser quality and quantity.
Oklahoma
Key pests of alfalfa in Oklahoma are the alfalfa weevil and an aphid complex. Alfalfa needs to be monitored throughout the growing season to be able to manage these and other alfalfa pests. Alfalfa weevil is a chronic problem in Oklahoma. The important biological control agent M. aethiopoides occurs only sporadically in Oklahoma. Dry weather conditions frequently coincide with peak alfalfa weevil occurrence, thus epizootics caused by Z. phytonomi are rare. Insecticides are the major management tool for alfalfa weevil. To properly time insecticide applications, a degree-day model based on accumulated degree-days and crop development is used. A network of weather stations are used to calculate growing F degree-days across Oklahoma, and degree-day accumulations made available to producers to use in making management decisions.
Blue alfalfa aphid, spotted alfalfa aphid, and pea aphid are also of importance in Oklahoma. Parasitoids effective against these pests rarely occur. The dry growing conditions in spring discourage the development of fungal pathogens. Host plant resistance was the most important management tactic for blue alfalfa aphid and spotted alfalfa aphid. Recently, a biotype of blue alfalfa aphid has emerged that has overcome existing plant resistance. This biotype is very virulent, and, if not properly managed, can kill alfalfa plants and result in loss of an entire alfalfa stand. Cultivars in Oklahoma are typically non-dormant types and will grow whenever temperatures permit. Spotted alfalfa aphid is a pest of alfalfa in winter. Spotted alfalfa aphid needs to be monitored on these winter stands. Spotted-alfalfa aphid resistant cultivars are available.
A complex of lepidopteran pests can threaten alfalfa in summer and fall cuttings. Potato leafhopper usually arrives during first crop alfalfa and needs t o be monitored on all crops.
Blister beetles, Epicauta spp. (Coleoptera: Meloidae), may be crushed when hay is crimped and conditioned, and then later trapped in bales. This is a concern because a natural toxin in these beetles, cantharidin, can kill livestock. Horses are the most susceptible animals, but cattle and sheep have also been poisoned. Blister beetles pass part of their life cycle as parasites of grasshoppers. Blister beetle outbreaks are therefore often tied to grasshopper outbreaks.
References
- Radcliffe, E. B. and K. L. Flanders. 1998. Biological Control of Alfalfa Weevil in North America. Integrated Pest Management Reviews 3: 225-249. Note: To access this paper as a PDF file on-line, you, or your organization, must be a subscriber to Integrated Pest Management Reviews.
- Frisbie, R. E. and P. L. Adkisson. 1985. CIPM: Integrated Pest Management on Major Agricultural Systems, from a symposium sponsored by The Consortium got Integrated Pest Management and USDA/CSRS. Texas Agr. Exp. Sta. MP-1616, 743 pp.
- Pass, B. C. A summary of CIPM research achievement in the alfalfa cropping system, pp. 16-34.
- Cuperus, G. W., E. B. Radcliffe, D. K. Barnes, and G. C. Marten. 1983. Economic injury levels and economic thresholds for potato leafhopper on alfalfa. J. Econ. Entomol. 76:1341-1349.
- Armbrust, E. J. and G. G. Gyrisco. 1982. Forage crops insect pest management. In: Introduction to Insect Pest Management, 2nd. edition, Metcalf, R. L. and W. H. Luckmann, editors. Chapter 11, pp. 443-463. Wiley-Interscience.
- Cuperus, G. W., E. G. Radcliffe, D. K. Barnes, and G. C. Marten. 1982. Economic injury levels and economic thresholds for pea aphid, Acyrthosiphon pisum (Harris), on alfalfa. Crop Protection 1: 453-463.
- Armbrust, E. J., B. C. Pass, D. W. Davis, R. G. Helgeson, G. R. Manglitz, R. L. Pienkowski, and C. G. Summers. 1980. General accomplishments toward better insect control in alfalfa. In: New Technology of Pest Control, C. B. Huffaker, editor. Wiley-Interscience, pp. 187-216.
- Kingsley, P. C., M. D. Bryan, W. H. Day, T. L. Burger, R. J. Dysart, and C. P. Schwalbe. 1993. Alfalfa weevil (Coleoptera: Curculionidae) biological control: Spreading the benefits. Environ. Entomol. 22:1234-1250.
- Manglitz, G. R. and R. H. Ratcliffe. 1988. Insects and mites. In Alfalfa and alfalfa improvement [eds. Hanson, A. A., D. K. Barnes, and R. R. Hill, Jr.], American Society of Agronomy, Inc., Agronomy Monograph no. 29.
- Blodgett, S. L. and R. A. Higgins. 1990. Blister beetles (Coleoptera: Meloidae) in Kansas alfalfa: influence of plant phenology and proximity to field edge. J. Econ. Entomol. 83: 1042-1048.